Aspiration Catheter Receives FDA Clearance
On March 17, the Irish medical technology company Perfuze announced that its Millipede88 aspiration catheter has received 510(k) clearance from the U.S. Food and Drug Administration (FDA).
This is Perfuze’s first aspiration catheter to enter the “super-bore” market, designed to simplify stroke thrombectomy procedures.

Millipede88's approval was mainly due to the positive results from the MARRS clinical study.
According to data released by Dr. Raul Nogueira of the University of Pittsburgh Medical Center (UPMC), the device achieved a delivery success rate of 96% in clinical trials; and in the treatment of M1 segment vessel occlusion, the first-pass recanalization success rate was 77%.
Dr. Nogueira stated that the catheter features a patented corrugated design, which maintains lumen shape in tortuous vessels and prevents the collapse commonly seen with traditional catheters, thereby ensuring stability during thrombectomy.

Perfuze states that the Millipede88 is the first 0.088-inch large-bore catheter on the market designed for direct aspiration. For physicians, this means that in some cases, using this device alone can achieve vascular recanalization, thereby shortening the procedure time and reducing the cost of instruments.
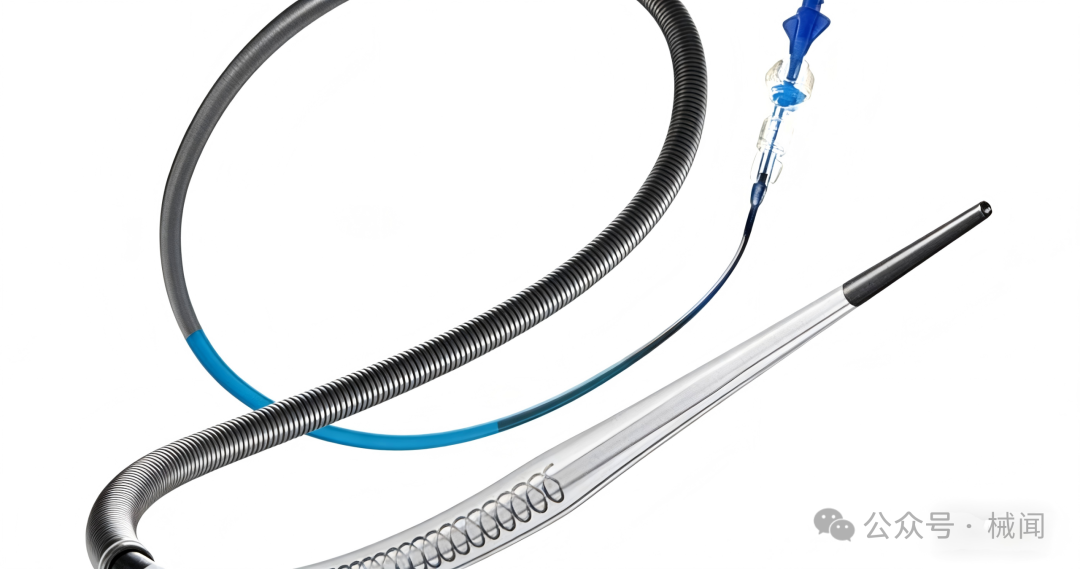
Perfuze CEO Wayne Allen said that Millipede88 will complement the company's previously approved Zipline vascular access catheter, providing support across all stages of neurovascular interventional procedures.
Perfuze plans to conduct a limited release in some comprehensive stroke centers in the United States, followed by a gradual expansion of market promotion.
【Copyright and Disclaimer】The above information is collected and organized by PlastMatch. The copyright belongs to the original author. This article is reprinted for the purpose of providing more information, and it does not imply that PlastMatch endorses the views expressed in the article or guarantees its accuracy. If there are any errors in the source attribution or if your legitimate rights have been infringed, please contact us, and we will promptly correct or remove the content. If other media, websites, or individuals use the aforementioned content, they must clearly indicate the original source and origin of the work and assume legal responsibility on their own.
Most Popular
-

Deadly Impact: Hormuz Strait Blockade Sparks Shortage of Plastic Raw Materials, Threatening Shutdowns at Japanese and Korean Chemical Plants
-

[Special Plastics Index] Geopolitical Conflicts Trigger Cost Surge, Plastics Futures and Spot Prices Rise Significantly Before Correction
-

Middle East Conflict Triggers "Plastic Rush": Zhangmutou Clogged With "Plastic Spring Festival Travel", Is This Frenzy Ultimately An Inventory Game?
-

Rolling to the material end! byd expands recycled plastic production by 100,000 tons amid market headwinds, redefining auto supply chain
-

Tao Lin: Tesla’s Supercharger Stations in China Surpass 2,500







