Zhejiang University Li Zhenglong's team: Photocatalytic oxidative depolymerization of polyethylene waste plastics into aliphatic long-chain dicarboxylic acids.
Recently, the global annual production of polyethylene (PE) has reached 83 million tons, making it one of the most widely used plastics. The long-term accumulation of PE waste poses a serious threat to the environment. This study demonstrates a method for the photocatalytic oxidation of polyethylene (PE) to generate higher-value aliphatic diacids (C4-C30) using a BiOI/BiVO4 p-n heterojunction material under visible light irradiation. This method achieves a high carbon yield of up to 83% under mild conditions, and by optimizing the composition of the heterojunction and reaction conditions, the yield of long-chain diacids (C10-C30) can be increased to 75%. The authors also explored the effects of reaction temperature, time, air pressure, and the stability of the catalyst after multiple cycles on the photocatalytic oxidation of PE. Various techniques were employed to elucidate the reaction mechanism: superoxide radicals (•O2-) are responsible for activating the C-H bonds in the PE main chain, while hydroxyl radicals (•OH) cleave the C=C bonds. Furthermore, the photocatalytic upgrading process has shown significant efficiency for various commercial PEs used in daily life. This photocatalytic system offers a promising approach for the efficient recycling of PE waste.
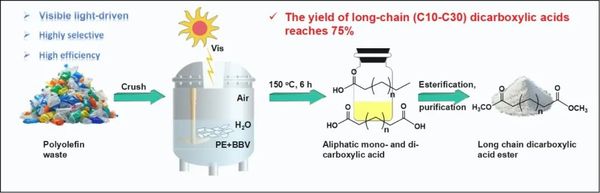
Figure 1. Schematic diagram of the photocatalytic oxidation depolymerization of polyethylene waste plastics to binary carboxylic acids proposed in this article.
Background introduction:
With the continuous increase in plastic waste, the efficient recycling of plastics has become a pressing environmental issue. Particularly for plastics like polyethylene (PE), their stability and durability result in extremely slow degradation rates in the environment, posing long-term threats to ecosystems. Currently, chemical recycling methods for plastics, especially polyethylene, face limitations such as high energy consumption and secondary pollution. To address these challenges, scientists are exploring more environmentally friendly and low-energy plastic recycling methods. Photocatalytic systems have emerged as an attractive option due to their use of renewable solar energy and relatively mild operating conditions. In recent years, photocatalytic systems have made progress in the degradation and recycling of plastic waste. Researchers aim to mimic natural degradation mechanisms through reactive species like free radicals, enabling the green recycling of polyolefin waste. The BiOI/BiVO4 p-n heterojunction material has been proposed as a photocatalyst due to its stability and non-toxicity. In this study, researchers developed this material and successfully achieved the photocatalytic oxidation of polyethylene under mild conditions using visible light irradiation. Against the backdrop of the search for eco-friendly, low-energy plastic recycling technologies, this study leverages the photocatalytic properties of the BiOI/BiVO4 p-n heterojunction material to efficiently convert polyethylene waste into higher-value chemicals.
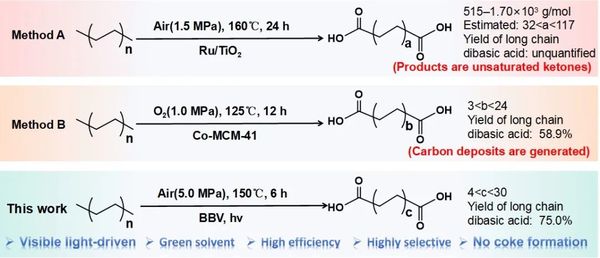
Figure 2. Comparison of this catalytic system with existing methods.
Highlights of this article:
This work successfully achieved the photocatalytic oxidation of polyethylene (PE) to valuable aliphatic dicarboxylic acids (C4-C30) under mild conditions using a BiOI/BiVO4 p-n heterojunction, with a carbon yield up to 83%. Notably, the carbon yield for long-chain dicarboxylic acids (C10-C30) reached 75%.
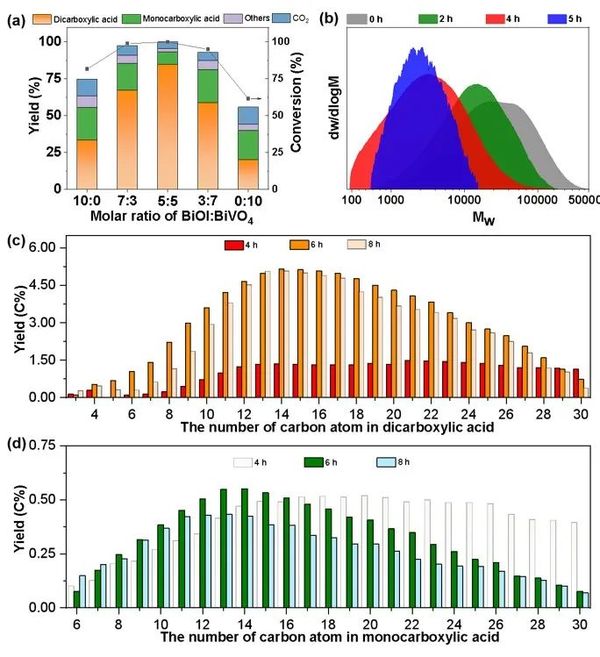
Figure 3. (a) Reaction results of PE on different catalysts, (b) Molecular weight of PE, (c) Dicarboxylic acids, (d) Distribution of monocarboxylic acid products over time.
In the photocatalytic oxidation process, superoxide radicals (•O₂⁻) and hydroxyl radicals (•OH) play a crucial role. The superoxide radicals activate the C-H bonds in the PE main chain, while the hydroxyl radicals oxidize and break the C=C bonds, ultimately converting PE into dicarboxylic acid products.
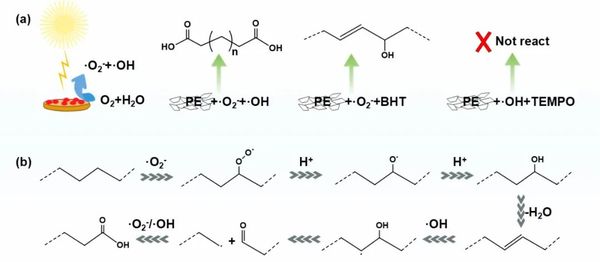
Figure 4. Proposed reaction mechanism
The catalytic system can oxidatively depolymerize most commercial polyethylene (PE) into dicarboxylic acids, with a carbon yield ranging from 59% to 64%. Trace amounts of branched alkanes and cycloalkanes were detected in the products. This photocatalytic system demonstrates excellent stability when processing commercial PE waste, indicating its potential for practical applications.
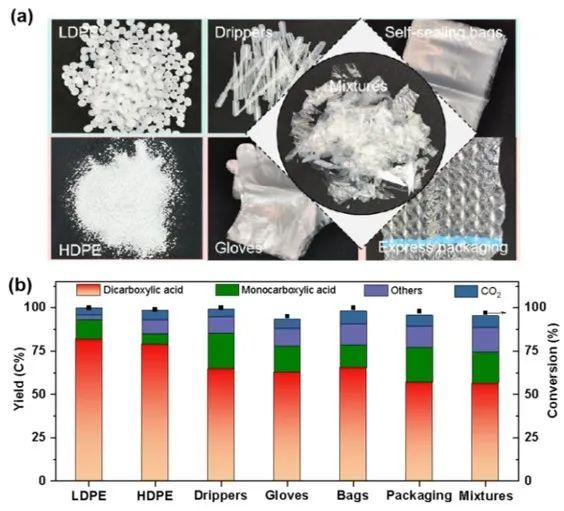
Figure 5. Results of photocatalytic oxidative depolymerization of pure PE particles and commonly found PE waste plastics in daily life.
Summary and Outlook:
Developing green chemical methods for the efficient recycling of waste polyolefins is crucial for reducing carbon emissions and supporting sustainable development. By converting PE into valuable chemicals (long-chain dicarboxylic acids), it is possible to add value to plastic waste, providing feedstock for the production of biodegradable polymers, lubricants, surfactants, and more. The photocatalytic oxidation method proposed in this study can be performed under mild conditions, significantly reducing the energy and resource inputs required by conventional methods, and is expected to advance the development of polyolefin waste plastic recycling technologies.
【Copyright and Disclaimer】The above information is collected and organized by PlastMatch. The copyright belongs to the original author. This article is reprinted for the purpose of providing more information, and it does not imply that PlastMatch endorses the views expressed in the article or guarantees its accuracy. If there are any errors in the source attribution or if your legitimate rights have been infringed, please contact us, and we will promptly correct or remove the content. If other media, websites, or individuals use the aforementioned content, they must clearly indicate the original source and origin of the work and assume legal responsibility on their own.
Most Popular
-

A Look at the Material Suppliers Behind SpaceX
-

Two Major Chemical Giants to Shut Down, Sell Again
-

BASF, Selling Again!
-

BASF Cuts Core Business As It Exits All Non-Core Operations, Triggering A New Round Of Industry Restructuring
-

Gas Explosion Accident at Shanxi Tongzhou Group Liushenyu Coal Mine Results in 82 Deaths







