Official document issued: Accelerated payment for consumables
The Terminator for Consumables Payment Dilemmas is Here?
01
Many places across the country
The healthcare insurance fund shifts from "post-payment" to "pre-payment".
In November last year, the National Medical Insurance Administration, in conjunction with the Ministry of Finance, issued the "Notice on Properly Carrying Out Medical Insurance Fund Prepayment Work," which clarified the connotation, conditions, and standards of the medical insurance fund prepayment policy, standardized the management of the prepayment process, accounting, and fund supervision, and pointed out that localities should implement the fund prepayment work according to the requirements of the notice.
The prepaid funds of the basic medical insurance fund are set up as working capital to help designated medical institutions alleviate the pressure of medical expense advances, improve medical service capabilities, and enhance the sense of gain for insured patients when seeking medical treatment. These funds are used for expenditures such as the procurement of medicines and medical consumables.
Medical institutions applying for advance payment must meet certain conditions.
The above document from Zhejiang clearly states that designated medical institutions within the coordinated area that meet the following basic conditions can apply for prepayment funds:

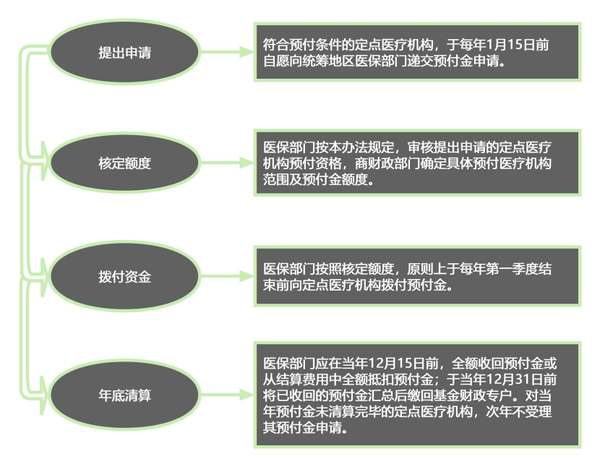

According to the Notice, a prepayment fund equivalent to 1 month will be allocated to designated medical institutions that meet the prepayment criteria. For designated medical institutions in the same coordinated area where the growth rate of basic medical insurance fund expenditures for the previous year was ≤5%, the comprehensive settlement or medical insurance performance assessment results for the most recent year were excellent, and they performed outstandingly in key medical insurance tasks, local areas may appropriately increase the scale of the prepayment fund based on actual circumstances, but it shall not exceed 2 months.
According to statistics, as of now, many regions such as Hunan, Yunnan, Zhejiang, Hubei, Inner Mongolia, Tibet, and Hebei have issued documents to promote the establishment of a prepaid fund management system. Previously, Guizhou, Heilongjiang and other places have also publicly solicited opinions on the management measures for prepaid medical insurance funds.
The shift in medical insurance fund settlement from a "post-payment system" to a "pre-payment system" will significantly alleviate hospitals' financial burden of advance payments and improve fund turnover efficiency. Under this chain reaction in the settlement turnover process, upstream players in the industry such as pharmaceutical companies and medical supply manufacturers will also benefit, with the issue of triangular debt in payments expected to ease.
02
"Three Settlements" is fully promoted
Can the dilemma of material reimbursement be resolved?
The issue of reimbursement for consumables has always been difficult to resolve.
Previously, Saibailan Medical Devices conducted a small-scale survey on the payment cycle of medical consumables, with a total of 281 participants voting. The voting results showed that the payment situation for centralized procurement (CP) medical consumables was significantly better than that for non-CP medical consumables. Forty-nine percent of the participants indicated that the payment cycle for CP medical consumables could be controlled within half a year. However, 84 participants stated that the payment cycle for the medical consumables related to their business had extended to more than one year.
The payment cycle for non-centralized procurement medical consumables is even longer, typically ranging from 1 to 2 years. However, 13 participants reported that their non-centralized procurement consumables could be paid within 3 months.
It is not hard to see that there are significant differences in the repayment situations across various regions and medical institutions, with many areas still experiencing prolonged repayment times.
In 2025, the "three settlements" of medical insurance became a hot topic in the field of drug and device payments. The national and local levels are vigorously promoting new measures, which are expected to solve the issue of triangular debts in payments.
Since the beginning of the year, the National Healthcare Security Administration has intensively disclosed the progress of the "three settlements" healthcare reform across multiple regions in the country, including provinces such as Beijing, Henan, Hubei, Anhui, Jiangxi, and Hainan. Reform measures in cities like Xiamen (Fujian), Anyang (Henan), Zhengzhou (Henan), Yunfu (Guangdong), Hami (Xinjiang), Binzhou (Shandong), and Dalian (Liaoning) have also been shared as case examples.
The "three settlements" in medical insurance refer to the immediate settlement between the medical insurance fund and medical institutions, the direct settlement between the medical insurance fund and pharmaceutical enterprises, and the simultaneous settlement between the medical insurance fund and commercial insurance, etc.
Previously, local governments have made many explorations in direct settlement. With the acceleration of instant settlement and synchronous settlement, the payment for consumables is expected to be fully expedited.
On March 26, the National Healthcare Security Administration disclosed the progress of the "three settlement" healthcare reform in Anyang, Henan. The report indicated that the region has preliminarily achieved the coordinated goal of "real-time settlement, direct settlement, and simultaneous settlement" across the entire healthcare fund chain.
In terms of instant settlement, the reimbursement time limit for medical insurance funds has been compressed from 30 working days after the designated medical institution's declaration to as short as 1 working day, with the first batch of 16 medical institutions included in the pilot program; direct settlement has expanded from 1 designated medical institution to 14, and the payment cycle for pharmaceutical distribution companies has been shortened to 25 days; a "one-stop" service window has been established in 5 pilot hospitals, achieving synchronous settlement for basic medical insurance and "Anhui Bao."
In terms of timing, the National Medical Insurance Administration has made it clear that by 2025, the nationwide basic implementation of direct settlement for centrally procured medicines and consumables, as well as national negotiation medicines, will be achieved. By 2026, direct settlement for these will be fully realized nationwide. Additionally, by 2025, about 80% of the coordinated regions will basically achieve instant settlement of basic medical insurance funds with designated medical institutions, and by 2026, all coordinated regions will realize instant settlement of basic medical insurance funds with designated medical institutions.
【Copyright and Disclaimer】The above information is collected and organized by PlastMatch. The copyright belongs to the original author. This article is reprinted for the purpose of providing more information, and it does not imply that PlastMatch endorses the views expressed in the article or guarantees its accuracy. If there are any errors in the source attribution or if your legitimate rights have been infringed, please contact us, and we will promptly correct or remove the content. If other media, websites, or individuals use the aforementioned content, they must clearly indicate the original source and origin of the work and assume legal responsibility on their own.
Most Popular
-

A Look at the Material Suppliers Behind SpaceX
-

BASF, Selling Again!
-

BASF Cuts Core Business As It Exits All Non-Core Operations, Triggering A New Round Of Industry Restructuring
-

Gas Explosion Accident at Shanxi Tongzhou Group Liushenyu Coal Mine Results in 82 Deaths
-

Bola Carbon Black Announces Global Restructuring of Specialty Carbon Black and Multi-Walled Carbon Nanotube Business







