New Study: SaBOX/Co Catalyst for the Facile Synthesis of Challenging Methacrylate-Vinyl Ester Copolymers
Copolymers synthesized from highly active and low active monomers face great challenges due to the significant differences in their reactivity and polymerization behavior.
The existing semi-batch/semi-continuous feeding strategy can synthesize certain copolymers but requires precise control of the monomer feeding process. For monomers with significantly different reactivities (copolymerization rates) such as methyl methacrylate (MMA) and vinyl acetate (VAc), it is easier to produce copolymers with highly uneven compositional distribution, or even mixtures containing homopolymers.
Although controlled radical polymerization (CRP) enables the construction of precise macromolecular structures, existing systems (such as RAFT and ATRP) cannot efficiently copolymerize vinyl esters and methacrylates directly, because almost all CRP systems can only control the polymerization of one type of vinyl monomer (either highly activated or less activated).
Even the cumbersome CRP techniques, stimulus-responsive RAFT switching, or post-modification and other indirect methods can only synthesize relatively simple diblock copolymers, which typically have low synthesis efficiency and are difficult to prepare multi-block or homogeneous random copolymers. Therefore, the straightforward synthesis of random copolymers and block copolymers of vinyl esters and methacrylates has been rarely successful to date, and achieving precise control of their structures is even more challenging.
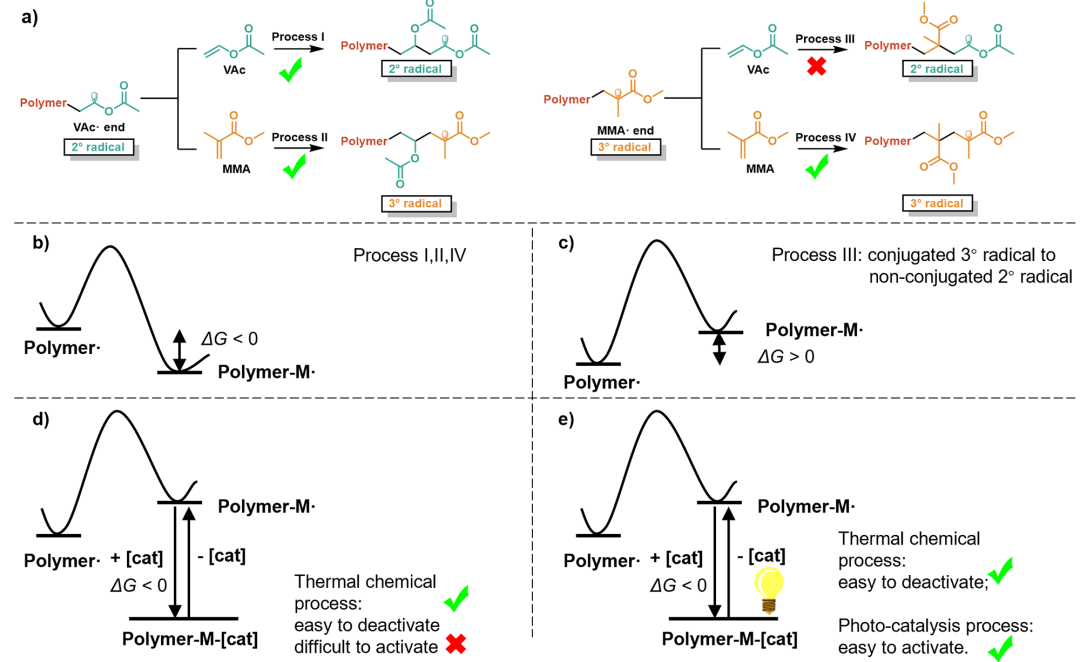
Figure 1 Analysis of VAc/MMA Copolymerization Process
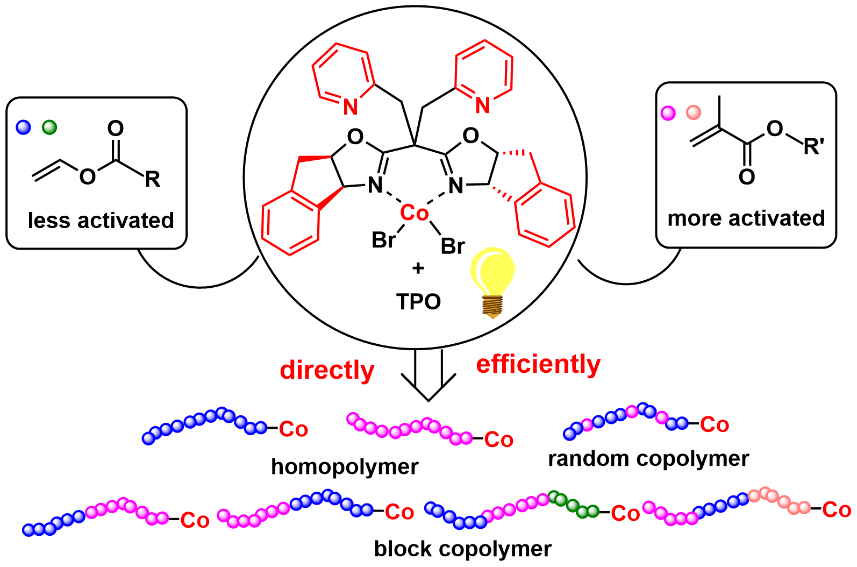
Figure 2 Simple synthesis of methyl acrylate-vinyl ester copolymer regulated by SaBOX/Co catalyst
In recent years, the team of Tang Yong/Wang Xiaoyan from the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, has conducted a series of studies on the design of novel controllable radical polymerization catalysts and the precise regulation of polymer structures.
Recently, the team analyzed four radical addition processes of copolymerization involving MMA and VAc monomers, and found that the addition of MMA radicals to VAc monomers involves the transformation from a stable conjugated tertiary carbon radical to a reactive non-conjugated secondary carbon radical. This process is thermodynamically highly unfavorable and prone to reverse reactions, posing a major obstacle to the copolymerization process.
The strategy of this team is to employ catalysts with strong binding (passivation) abilities to capture highly reactive secondary carbon radicals and generate dormant species, thereby enhancing their thermodynamic stability and preventing reverse reactions, making this process feasible. The team designed a bis(oxazoline)/CoBr₂ catalyst featuring an indene backbone and pyridine side arms for photoinitiated cobalt-mediated radical polymerization (CMRP). This catalyst provides a shielded coordination environment that avoids the influence of solvent and polar monomer groups on coordination. Compared with traditional CMRP catalysts, it exhibits stronger and differentiated binding (passivation) abilities toward various growing chain radicals, which not only prevents radical side reactions but also modulates the radical addition process during the copolymerization of high- and low-activity monomers, thereby narrowing the difference in reactivity ratios between the two types of monomers. This approach enables, for the first time, the straightforward and direct synthesis of di- and triblock copolymers of vinyl esters and methacrylates, as well as random copolymers with uniform component distribution. In-depth mechanistic studies are consistent with the polymerization results, validating the CMRP mechanism and the catalyst design concept.
This study successfully achieved the facile synthesis of copolymers from two types of monomers with significantly different reactivity by rationally designing catalysts. This breakthrough could contribute to the development of high-performance catalysts and polymer materials.
The work titled "A SaBOX/Co catalyst designed for facile access to the challenging copolymers of vinyl ester and methacrylate" was published in Nature Communications. The corresponding authors are Associate Researcher Wang Xiaoyan and Researcher Tang Yong from the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences. The first author is Ding Yijie, a doctoral student jointly trained by the Shanghai Institute of Organic Chemistry and ShanghaiTech University. This research was supported by the National Natural Science Foundation of China, the Strategic Priority Research Program of the Chinese Academy of Sciences, and the Science and Technology Commission of Shanghai Municipality.
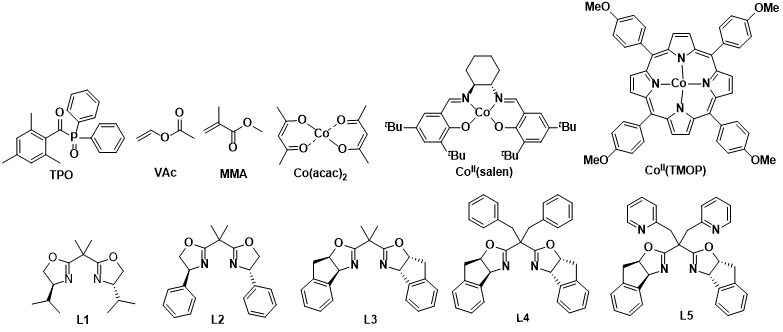
Figure 3. Structures of ligands, catalysts, initiators, and monomers
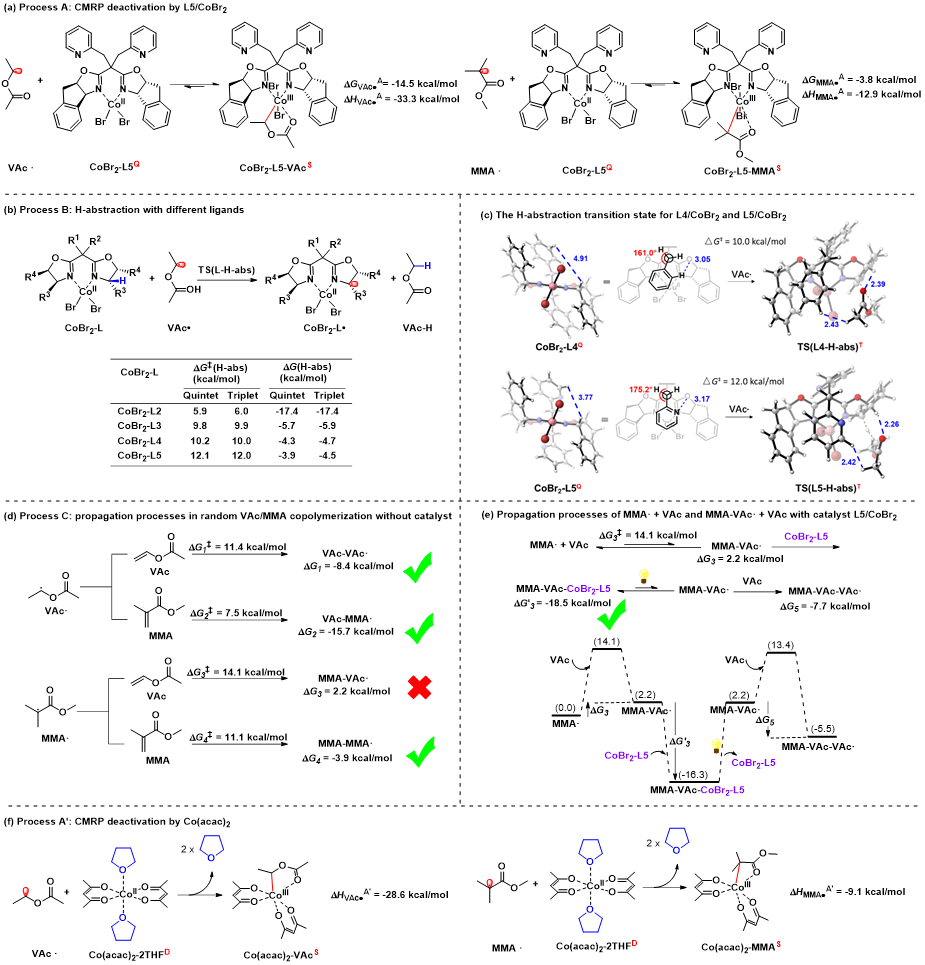
Figure 4 DFT Calculation
【Copyright and Disclaimer】The above information is collected and organized by PlastMatch. The copyright belongs to the original author. This article is reprinted for the purpose of providing more information, and it does not imply that PlastMatch endorses the views expressed in the article or guarantees its accuracy. If there are any errors in the source attribution or if your legitimate rights have been infringed, please contact us, and we will promptly correct or remove the content. If other media, websites, or individuals use the aforementioned content, they must clearly indicate the original source and origin of the work and assume legal responsibility on their own.
Most Popular
-

A Look at the Material Suppliers Behind SpaceX
-

Two Major Chemical Giants to Shut Down, Sell Again
-

BASF, Selling Again!
-

BASF Cuts Core Business As It Exits All Non-Core Operations, Triggering A New Round Of Industry Restructuring
-

Gas Explosion Accident at Shanxi Tongzhou Group Liushenyu Coal Mine Results in 82 Deaths







