Medtech home: Top 10 Innovative Implantable and Interventional Products of 2026
Over the past year, China’s core medical device sector—implantable and interventional devices—has entered a golden era of high-quality development, driven by technological innovation and grounded in clinical value.
Since its establishment over a decade ago, DeviceHome has remained deeply committed to China's interventional and implantable medical device sector, witnessing and documenting every milestone in this field. Based on thousands of industry reports published in 2025, we focused on three core dimensions—"Breakthrough Technological Innovation," "Clinical Application Suitability," and "Industry Ecosystem Impact"—and, through rigorous selection incorporating authoritative expert reviews and industry reader feedback, have ultimately identified the ten most benchmark-setting interventional and implantable innovative products of the year. We proudly unveil the "DeviceHome: Top 10 Interventional & Implantable Innovative Products of 2026" list.
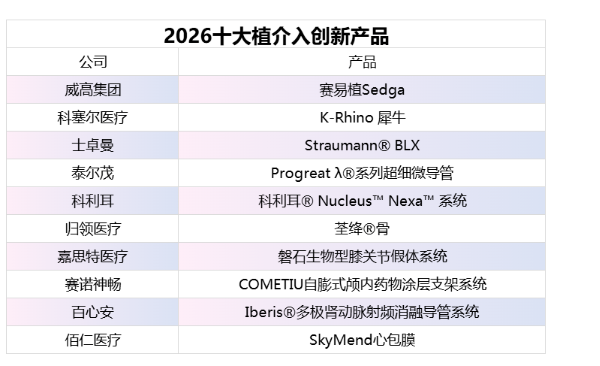
*The above list is ranked without any order.
Inclusion reason

Product Introduction
Inclusion reason
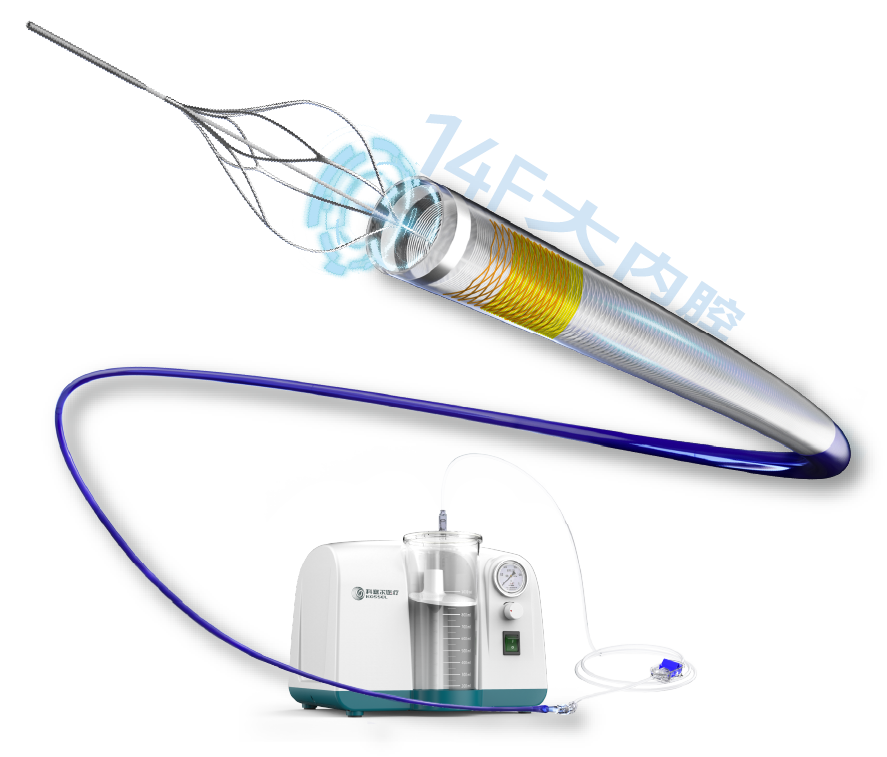
K-Rhino Rhinoceros™ 14F large-bore aspiration catheter has outstanding clearance efficiency, fast speed, and high removal rate. Thanks to the large-diameter design, large-bore aspiration reduces the need for repeated operations, and the amount of blood loss during surgery is controllable. With the 14F specification, effective hemostasis can be achieved with routine postoperative compression, offering good safety.
Product Introduction
K-Rhino Peripheral Blood Clot Aspiration System, featuring a 14F large lumen aspiration, with four clinical advantages:
-
14F ultra-large inner diameter, 1.4× enhanced thrombus removal force, significantly shortened aspiration time, reducing procedure duration.
-
Large-lumen aspiration reduces repeated manipulations and significantly lowers total blood loss compared to conventional 8F, 10F, and 12F catheters, enhancing procedural safety.
-
14F puncture site management is well-established; popliteal and femoral vein punctures are safe and effective, and routine compression is sufficient for hemostasis.
-
Large-bore design copes with low-pressure environments, ensuring stable suction efficiency, suitable for diverse surgical scenarios.
Reason for Selection
Guided by the principles of aesthetic restoration, the Straumann® BLX implant system features a unified connection, with streamlined transmucosal contours in its prosthetic components. The entire product line is streamlined yet remains versatile and fully functional.

Product Introduction
Straumann® BLX, designed for immediate implantation with dynamic bone management, enables minimally invasive and predictable immediate implant solutions in all types of alveolar bone. This implant system upholds the high standards and precision of Swiss quality, integrating pioneering innovative designs backed by long-term scientific evidence, truly allowing dental professionals to carry out treatments with peace of mind, reassurance, and confidence.
Reason for Selection
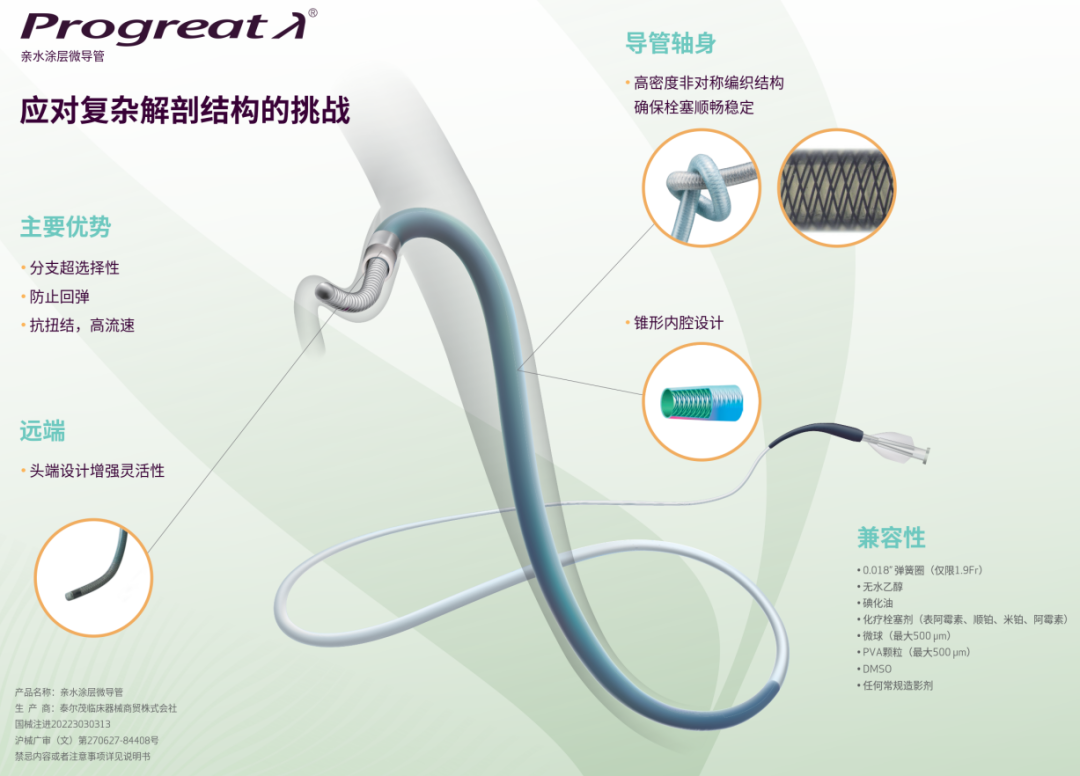
The technical breakthrough of the Progreat λ® series microcatheters marks the official entry of tumor interventional therapy into an era of refinement and personalization, opening up a completely new technological pathway for enhancing the interventional treatment outcomes of liver cancer and other tumors.
Product Introduction
This series of microcatheters has achieved significant innovation in core technology: the ultra-fine lumen allows them to easily reach small tumor feeding vessels that traditional catheters cannot access; excellent trackability ensures smooth navigation through tortuous vessels; stable positioning provides a reliable guarantee for precise treatment. Particularly noteworthy is its unique variable diameter design, which maintains the ultra-fine characteristics at the tip while ensuring overall stability of the catheter, effectively preventing tip displacement under high-pressure injection.
In clinical applications, Progreat λ® not only demonstrates excellent high-pressure resistance and compatibility with embolic materials, but also shows precise super-selective capabilities in handling complex cases such as ectopic renal artery supply. It particularly excels in comprehensive treatment plans involving detailed TACE combined with particle implantation, immunotherapy, and more.
Reason for Selection
Nucleus™ Nexa™ System is the world's first intelligent cochlear implant system to apply advanced intelligence, and it features a chip with powerful computing and storage capabilities.
Product Introduction
The new Nucleus Nexa system integrates more than 10 years of R&D achievements, for the first time applying an intelligent chip with high computing and storage capabilities to the Nexa implant, marking a breakthrough technological innovation in the cochlear implant industry in nearly 20 years.
Guiling Medical: Quanjiang® Bone
Inclusion reason
Quanjiang® Calcium Sulfate Bone is the first and only calcium sulfate artificial bone product in China that has been approved through large-scale clinical trials. Compared with imported similar calcium sulfate products, it demonstrates more excellent clinical performance, and its accompanying tools are more complete and comprehensive. It is expected to become a key force in achieving import substitution under the national centralized procurement context.
Product Introduction
Quanjiang® bone is primarily composed of high-purity α hemihydrate calcium sulfate, with a purity exceeding 98%. Its unique crystal structure design enables a compressive strength more than 6 times that of human cancellous bone, and over 2 times that of similar imported products. When implanted into human bone defect areas, it can serve as a scaffold material, promoting bone repair and regeneration, and meeting the clinical needs of various bone repair subfields, including trauma, joint, spine, and bone tumors.
The product exhibits excellent biocompatibility and can be fully degraded and absorbed by the surrounding tissues within the patient’s body, achieving a degradation period of 2–3 months, with degradation kinetics closely matching the bone regeneration rate. Through process optimization of the material’s pore structure, antibiotics, antitumor agents, antitubercular drugs, and other therapeutics can be quantitatively loaded, enabling high-concentration, sustained release over 5–6 weeks. This achieves the clinical efficacy of localized drug delivery, reduces adverse reactions, improves drug utilization, and enhances therapeutic outcomes.
Jiaste: Panstone Biologic Knee Prosthesis System
Reason for Selection
Compared with traditional cemented prostheses, biological knee prostheses eliminate the risk of cement toxicity and allergic reactions, effectively reduce the incidence of venous thrombosis and aseptic loosening, and avoid thermal injury to soft tissues, providing patients with a more stable and reliable postoperative recovery.

Product Introduction
Selection Criteria
The COMETIU stent system is the world’s first self-expanding, nitinol drug-coated stent system specifically designed for intracranial atherosclerotic stenosis (ICAS) and delivered via microcatheter. It is also the first domestically developed neurointerventional device to receive U.S. FDA Breakthrough Device designation.

Product Introduction
In August 2025, SINOMED’s subsidiary SINOMED Neuro received FDA Breakthrough Device Designation for its self-expanding intracranial drug-coated stent system, COMETIU, independently developed by SINOMED Neuro.
COMETIU is a closed-loop, recyclable stent with excellent vessel wall apposition, avoiding the issue of in-stent restenosis caused by inward curling of open-loop stents into the vascular lumen in cases of significant residual stenosis. Its patented eG™ drug-eluting coating enables optimal drug release post-implantation: first, it ensures arterial wall drug concentrations align precisely with the timeline of smooth muscle cell proliferation, thereby effectively and selectively inhibiting excessive smooth muscle cell proliferation; second, it achieves zero drug residue after 30 days, minimizing drug-induced inhibition of endothelial cells and thereby promoting endothelial healing. Furthermore, its neurotoxicity has been confirmed safe through animal studies.
Reason for Selection
An Tong Medical (Bai Xin An Group)'s independently developed Iberis® multi-electrode renal artery radiofrequency ablation catheter system is the world's only RDN product that can achieve dual access via the radial and femoral arteries.

Product Introduction
The Iberis® Multi-electrode Renal Artery Radiofrequency Ablation Catheter System employs advanced renal nerve ablation technology. Through a minimally invasive interventional approach, the ablation catheter is precisely delivered to the renal artery, where radiofrequency energy is applied to thoroughly ablate the renal sympathetic nerves, thereby interrupting excessive renal sympathetic nerve signal transmission and offering new hope and treatment options for patients with hypertension.
Inclusion reason
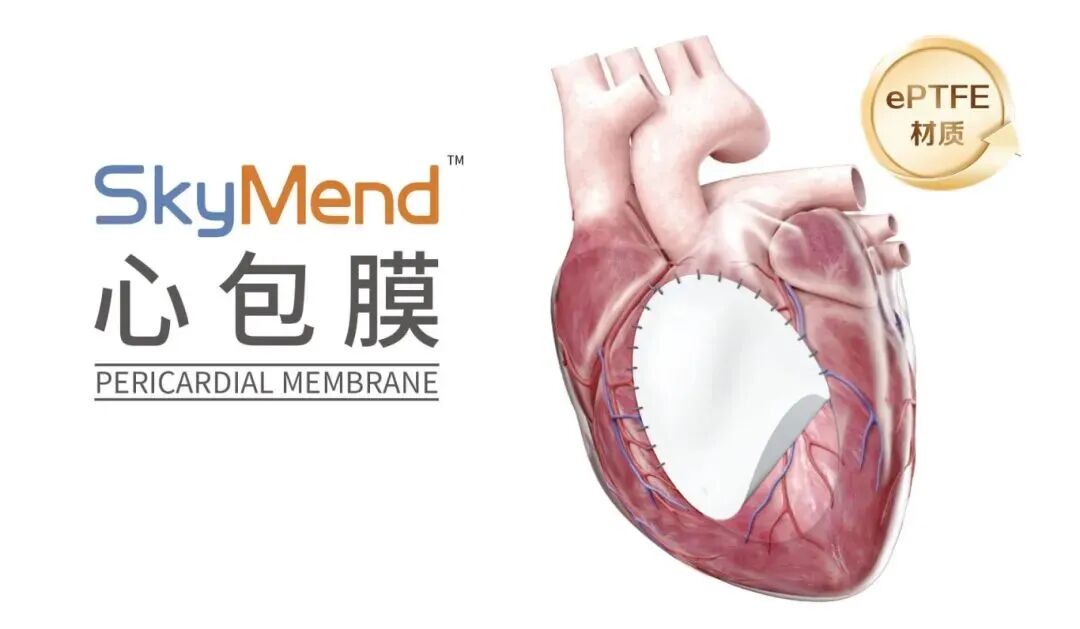
SkyMend Pericardial Membrane is used for pericardial membrane reconstruction or repair. As China's first ePTFE (expanded polytetrafluoroethylene) patch product, it fills the gap in domestic research and production technology of ePTFE materials, and has the potential to address the long-term domestic monopoly of imported ePTFE implantable medical devices and materials.
Product Introduction
【Copyright and Disclaimer】The above information is collected and organized by PlastMatch. The copyright belongs to the original author. This article is reprinted for the purpose of providing more information, and it does not imply that PlastMatch endorses the views expressed in the article or guarantees its accuracy. If there are any errors in the source attribution or if your legitimate rights have been infringed, please contact us, and we will promptly correct or remove the content. If other media, websites, or individuals use the aforementioned content, they must clearly indicate the original source and origin of the work and assume legal responsibility on their own.
Most Popular
-

Continental Plans to Begin Sale of ContiTech in Early 2026
-

$4 Billion! Medtronic Makes Another Acquisition
-

BASF Delivers First Batch of Innovative Cathode Materials for Semi-Solid-State Batteries to Weilan New Energy
-
Profit and Revenue Growth Struggle to Conceal Debt Repayment Pressure; Success of Kingfa Sci & Tech's High-End Strategy Yet to Be Seen
-

Why did a century-old european dental instrument giant relocate its manufacturing hub to china?