Latest technological evolution, market landscape, and future trends of nylon flame retardants
The nylon flame retardant industry is currently at a critical juncture of technological paradigm shift—on one hand, regulations such as the EU REACH continue to tighten restrictions on the use of halogenated flame retardants; on the other hand, emerging applications like new energy vehicles and ultra-thin electronic devices are demanding higher flame retardancy efficiency and better retention of mechanical properties.
Nylon, as the top of the five engineering plastics, has inherent flammability that severely limits its application in high-safety-demand fields such as electronics and electrical, automotive, and aerospace. With the tightening of global environmental regulations (such as EU REACH, RoHS) and the improvement of performance requirements from downstream industries, flame retardant technology is undergoing a profound transformation, shifting from halogenated to halogen-free, from additive to reactive, and from single-function to multi-functional synergy.
This article, by reviewing the flame-retardant mechanisms of nylon, mainstream technological approaches, industrial landscape, and recent research advances, reveals the core industry challenge of balancing flame retardancy and mechanical properties, and identifies interfacial engineering, nanocomposite technologies, and reactive flame retardants as key directions for future breakthroughs.

Overview and Market Landscape of Flame Retardant Nylon Industry
1. The Necessity of Flame Retardant Nylon
Nylon (PA6, PA66, etc.) is widely used in the textile, automotive, and electrical and electronic fields due to its excellent mechanical properties, wear resistance, and chemical corrosion resistance. However, nylon is inherently flammable, with a Limiting Oxygen Index (LOI) of only 21%-24%, and a UL94 flammability rating typically at V-2. During combustion, it produces a large amount of molten droplets, which can easily spread the flame, leading to a secondary fire risk. Especially in the glass fiber reinforced nylon (GF/PA) system, the "wick effect" further accelerates flame propagation, making flame retardant modification an inevitable choice for its high-end applications.
2. Global and Chinese Market Size
According to the latest research data from QYResearch and Global Info Research, the global flame-retardant nylon market is demonstrating steady growth.
Market SizeIn 2025, the global flame-retardant nylon market size is estimated to be approximately RMB 8.418 billion to RMB 11.86 billion (around USD 1.2–1.3 billion), and is projected to grow to RMB 14.53 billion to RMB 20.26 billion by 2031–2032, maintaining a compound annual growth rate (CAGR) of8.0%-8.2%between
Regional PatternChina is the world's largest producer of flame-retardant nylon (accounting for about 65%) and the largest consumption market (accounting for about 46%), followed by Europe and North America. This situation is closely related to China's status as the global manufacturing hub and its large demand from the electronics and electrical, and automotive industries.
3. Competitive Ecosystem
The global flame-retardant nylon market exhibits a relatively high concentration, with key players including international chemical giants such as DuPont, DSM, Celanese, and BASF, as well as leading domestic enterprises like Kingfa Science & Technology and BenSong New Materials. The top five manufacturers collectively account for approximately 52% of the global market share. Domestic enterprises’ rapid advancement in the halogen-free flame-retardant segment is gradually reshaping the high-end market landscape, which has historically been dominated by foreign companies.
Flame Retardant Mechanism and Technical Route of Nylon
1. Combustion and Flame-Retardant Mechanisms
The burning of nylon is a complex free radical chain reaction involving thermal-oxidative degradation, producing flammable gases (such as hydrocarbons), heat, and soot. The mechanisms of action for flame retardants mainly fall into the following categories:
Gas-phase flame retardancyFlame retardants decompose to generate inert gases (e.g., N₂, NH₃, H₂O), diluting the concentrations of oxygen and combustible gases; or release radical scavengers (e.g., halogen- or phosphorus-containing radicals) to interrupt the combustion chain reaction.
Condensed-phase flame retardancyPromotes the formation of a dense char layer on the burning surface of nylon. This char layer acts as a physical barrier, isolating heat, oxygen, and flammable gases, while also suppressing melt dripping.
Interrupt heat exchangeBy promoting molten droplets to carry away heat, the material temperature is reduced below its ignition point. However, pure molten droplets need to be carefully controlled to prevent the droplets from igniting other objects.
2. Core Flame-Retardant Technology System
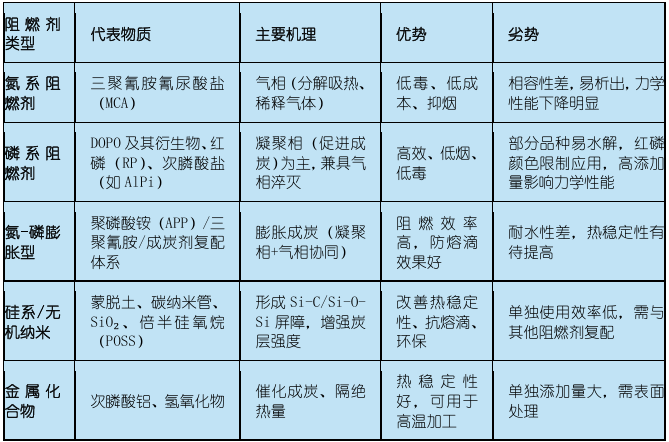
Halogen-containing flame retardant system
Halogenated flame retardantsBrominated flame retardants (such as polybrominated diphenyl ethers) have high flame retardant efficiency, require small addition amounts, and have minimal impact on the mechanical properties of materials. However, they release corrosive halogenated hydrogen gases and highly toxic dioxins during combustion, causing "secondary pollution." Although halogen-containing systems still account for approximately 51% of the market share, they are being strongly driven by environmental regulations (such as the EU RoHS directive and REACH regulation) to be replaced.Halogen-free has become an irreversible trend.
(2) Halogen-free flame retardant system
Halogen-free flame retardants are currently the core focus of research, development, and application, primarily including the following types:
Frontier Technological Breakthroughs and Scientific Research Progress (2025–2026)
Recently, both academia and industry have made significant breakthroughs in addressing the core challenge of balancing flame retardancy and mechanical properties, primarily exhibiting two major trends:Multifunctionality and Interface Synergistic Design、Reactive Intrinsic Flame Retardancy。
Temperature-responsive Dual-functional Interface Design
For glass fiber-reinforced nylon (GF/PA), the “wick effect” poses a significant challenge to flame retardancy, and conventional approaches often result in trade-offs.
Breakthrough achievementA joint team from Chongqing Jiaotong University and the IMDEA Materials Institute in Spain has developed aTemperature-responsive Iron/Phosphorus Triazine Complex Silane Coupling Agent (APETP-Fe)。
Collaboration Mechanism:
→ Processing StageAt a processing temperature of approximately 235 °C, the active hydroxyl groups of this molecule undergo dynamic transesterification reactions with the amide groups of nylon 6, forming bonds between the fiber and the matrix.High-strength covalent bondAchieve molecular-level interfacial reinforcement, significantly enhancing mechanical properties.
→ Combustion PhaseWhen exposed to high temperatures, the coupling agent triggers an interfacial catalytic charring mechanism. Iron ions and the cyclotriphosphazene coordination structure exert a super-efficient catalytic effect, generating a continuous and dense char layer in situ on the surface of the fibers, physically isolating oxygen and cutting off the molten flow path, thereby inhibiting the "wick effect."
This "high-temperature catalytic charring, low-temperature bond strengthening" strategy successfully enhances both the flame retardancy and mechanical strength of the composite material, providing a new paradigm for the design of high-performance fiber-reinforced composites.
Reactive flame retardant (intrinsic flame retardant nylon)
Although additive flame retardants offer simple processing, they commonly suffer from issues such as migration and blooming, as well as poor compatibility. A research team from Nanjing University of Aeronautics and Astronautics has proposed an alternative solution—Reactive Flame Retardant。
Technical CoreFlame retardants containing dicarboxyl groups (BNPPO) and diamine groups (PDPPD) were synthesized, and phosphorus (P) and nitrogen (N) flame-retardant elements were directly introduced into the main chain of nylon 66 through copolymerization.
Flame-retardant elements are chemically bonded to the polymer molecular chain, fundamentally solving the migration problem. This method endows the material with excellent flame-retardant properties while maximizing the retention of the original mechanical and thermal properties of nylon, achieving "intrinsic flame retardancy."
3. Nano-composites and multi-element synergy
Simple single flame retardants often fail to meet stringent requirements, making multi-element (P-N-Si) synergy and nanocomposite technology the mainstream.
Layered Catalytic CharringResearch shows that by constructing a stacked structure such as ZrAA@Fe@ZIF-67, atomic or nanoscale catalytic carbonization can be achieved, greatly improving the compactness and thermal stability of the carbon layer.
Nanofiller SynergyBlending MCA with carbon black (CB), utilizing the dispersion effect of carbon black to improve the compatibility of MCA in nylon 6, thereby enhancing both flame retardancy and spinnability.
Challenges and Future Prospects
1. The core challenges currently faced
Environmental friendlinessNot only must the flame retardant itself be non-toxic, but it must also impose no environmental burden throughout its entire life cycle, including recycling and degradation. Attention should also be paid to the toxicity of combustion byproducts from certain halogen-free flame retardants and the use of solvents during their modification processes.
Cost pressureHigh-performance halogen-free flame retardants (such as structurally specific phosphinate salts and DOPO derivatives) are relatively expensive, limiting their widespread adoption in cost-sensitive applications.
The challenge of dispersing nanomaterialsNano flame retardants show significant effects, but they tend to agglomerate easily, and achieving uniform and stable nano-scale dispersion in polymer matrices still faces engineering challenges.
2. Future Development Trends
Intelligence and multifunctionalityFuture flame retardants will no longer be merely "additives," but rather part of smart materials. For example, temperature-responsive coupling agents can perform different functions according to environmental changes. Additionally, flame retardant materials that combine thermal conductivity, antibacterial, and antistatic properties will become research hotspots.
With the advancement of the “Dual Carbon” strategy, bio-based flame retardants derived from renewable resources (e.g., phytic acid, tannic acid) will gain increasing favor. Meanwhile, designing reversible crosslinked flame-retardant systems that are easy to recycle and suitable for closed-loop reuse is also a critical direction.
Digital Simulation and High-Throughput ComputingUsing artificial intelligence and molecular simulation technologies to predict the performance of flame retardants with different structures, accelerating the screening and development cycle of new flame retardants.
【Copyright and Disclaimer】The above information is collected and organized by PlastMatch. The copyright belongs to the original author. This article is reprinted for the purpose of providing more information, and it does not imply that PlastMatch endorses the views expressed in the article or guarantees its accuracy. If there are any errors in the source attribution or if your legitimate rights have been infringed, please contact us, and we will promptly correct or remove the content. If other media, websites, or individuals use the aforementioned content, they must clearly indicate the original source and origin of the work and assume legal responsibility on their own.
Most Popular
-

Continental Plans to Begin Sale of ContiTech in Early 2026
-

$4 Billion! Medtronic Makes Another Acquisition
-

BASF Delivers First Batch of Innovative Cathode Materials for Semi-Solid-State Batteries to Weilan New Energy
-
Profit and Revenue Growth Struggle to Conceal Debt Repayment Pressure; Success of Kingfa Sci & Tech's High-End Strategy Yet to Be Seen
-

Why did a century-old european dental instrument giant relocate its manufacturing hub to china?








