Approved by FDA! Novel Guiding Catheter for Stroke
Recently, the Irish company Perfuze, which specializes in developing thrombectomy devices for stroke patients, announced that its new guide catheter Zipline has been approved by the FDA for market release, and it has completed a financing of 22 million euros (approximately 170 million RMB).
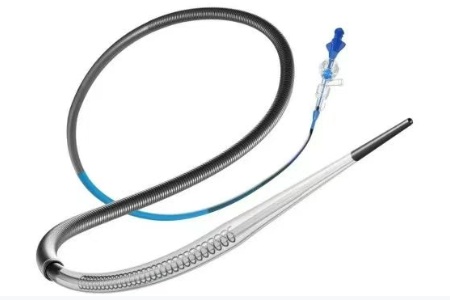
▲New Guide Catheter Zipline
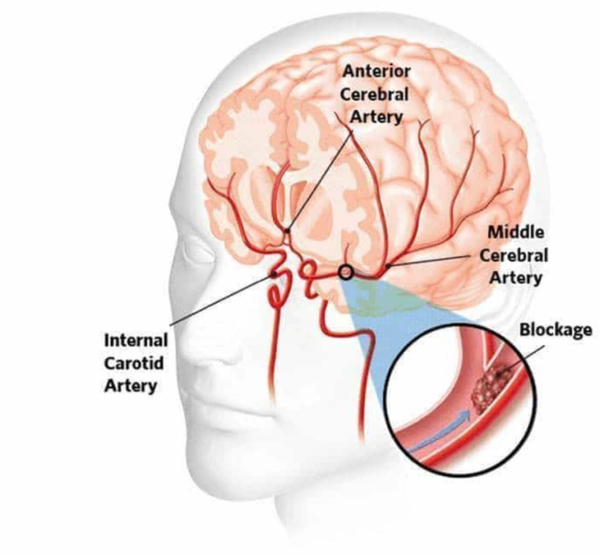
Stroke is a common and serious cerebrovascular disease, which refers to the occlusion of cerebral blood supply arteries due to the formation of atherosclerotic plaques, plaque rupture, thrombosis, or embolism, leading to ischemic and hypoxic necrosis of brain tissue, thereby causing neurological dysfunction.
This disease is characterized by high incidence, high disability rate, and high mortality worldwide, bringing a heavy burden to patients and their families. According to statistics, one quarter of adults over the age of 25 globally will experience a stroke in their lifetime. In the treatment of stroke, time is crucial, and rapid restoration of blood flow to the brain is key to improving patient outcomes.
In recent years, with the continuous advancement of medical technology, the treatment methods for stroke have also been continuously developing. Endovascular intervention, as an effective treatment method, has gradually become the focus of clinical attention. Among these, the guide catheter plays a crucial role in endovascular intervention. It can provide a stable delivery channel for interventional devices, helping doctors to more accurately reach the lesion site, thereby improving the success rate and safety of the surgery.
Zipline can significantly improve the tracking and delivery performance of large inner diameter (070) and ultra-large inner diameter (088) aspiration catheters, simplifying the neurointerventional procedure and achieving faster and more efficient stroke treatment.
By enhancing support and operational flexibility, Zipline is committed to optimizing thrombus removal efficiency, thereby improving surgical success rates and patient outcomes. FDA approval marks a significant step forward in Perfuze's mission to simplify and improve stroke treatment and outcomes through technological innovation.
# Executive and Expert Evaluations
Perfuze Chairman Hooman Hakami commented: "The continued support from investors demonstrates confidence in our technology and vision. This round of funding will help us to conduct limited market promotion in the US and advance the implementation of revolutionary stroke treatment solutions at selected centers."
"Zipline's FDA approval demonstrates our commitment to developing world-leading stroke solutions. This authorization will strengthen our presence in the U.S. market and drive us to provide truly innovative, efficient, and user-friendly technologies that transform the landscape of stroke treatment."
"Zipline catheter represents an innovative technology that I believe will simplify stroke intervention, reduce costs, and accelerate reperfusion. Based on my initial experience, they can achieve rapid clot access and aspiration even in complex anatomies," said Dr. Jay Dolia, Assistant Professor of Neurology at Emory University School of Medicine.
Perfuze is an Irish company that currently focuses on the development of ultra-large bore thrombectomy technology, also known as the Millipede catheter. The founders of the company are Wayne Allen and Liam Mullins.
The company has undergone three rounds of financing, and the known specific information is as follows:
-
On January 29, 2019, the company announced that it had raised 3 million euros in a seed funding round, led by EarlyBirdCapital, with participation from MedFocus Fund and Enterprise Ireland. -
On February 9, 2022, the company announced that it had raised 22.5 million euros in Series A financing, led by Seroba Life Sciences and EQT Life Science, with participation from SV Health Investors, Medtech Convergence Fund, LSP Health Economics Fund, HBM-MedFocus, and EarlyBird. -
In March 2025, the company announced the completion of a 22 million euro financing. This round of financing was led by existing investors (Earlybird, EQT Life Sciences, Seroba, and SV Health, among others). The funds will support the commercial promotion of Perfuze's two marketed products, the guide catheter Zipline and the large-bore aspiration catheter Millipede, in the United States, and advance the clinical progress of its pipeline products.
【Copyright and Disclaimer】The above information is collected and organized by PlastMatch. The copyright belongs to the original author. This article is reprinted for the purpose of providing more information, and it does not imply that PlastMatch endorses the views expressed in the article or guarantees its accuracy. If there are any errors in the source attribution or if your legitimate rights have been infringed, please contact us, and we will promptly correct or remove the content. If other media, websites, or individuals use the aforementioned content, they must clearly indicate the original source and origin of the work and assume legal responsibility on their own.
Most Popular
-

A Look at the Material Suppliers Behind SpaceX
-

Two Major Chemical Giants to Shut Down, Sell Again
-

BASF, Selling Again!
-

BASF Cuts Core Business As It Exits All Non-Core Operations, Triggering A New Round Of Industry Restructuring
-

Gas Explosion Accident at Shanxi Tongzhou Group Liushenyu Coal Mine Results in 82 Deaths







