Adjustment of Medical Insurance Consumables Directory Effective February 10 (List Attached)
The management of consumable payments is entering a new phase, with national coordination beginning to bear fruit.
Fujian Launches First 2026 Catalog Adjustment
Effective February 10th
February 10th, Fujian Medical Insurance Bureau issued the "Notice on the First Dynamic Adjustment of the <Fujian Province Medical Insurance Medical Consumables Payment Management Directory (Trial Implementation)>" (attached at the end of the article).


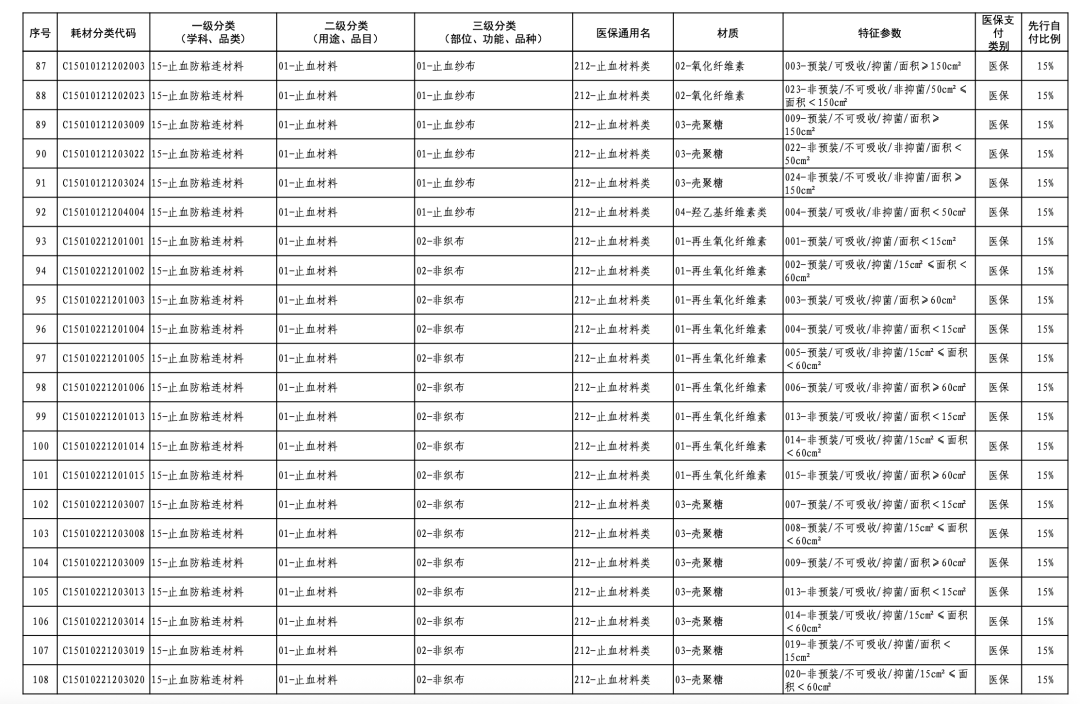
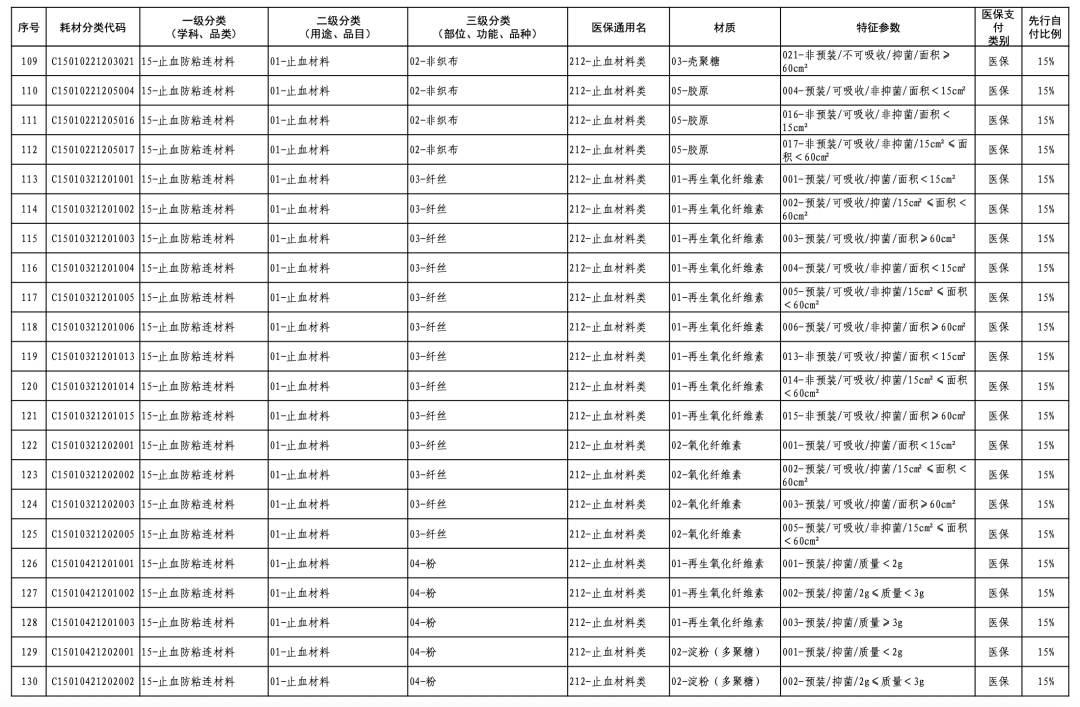
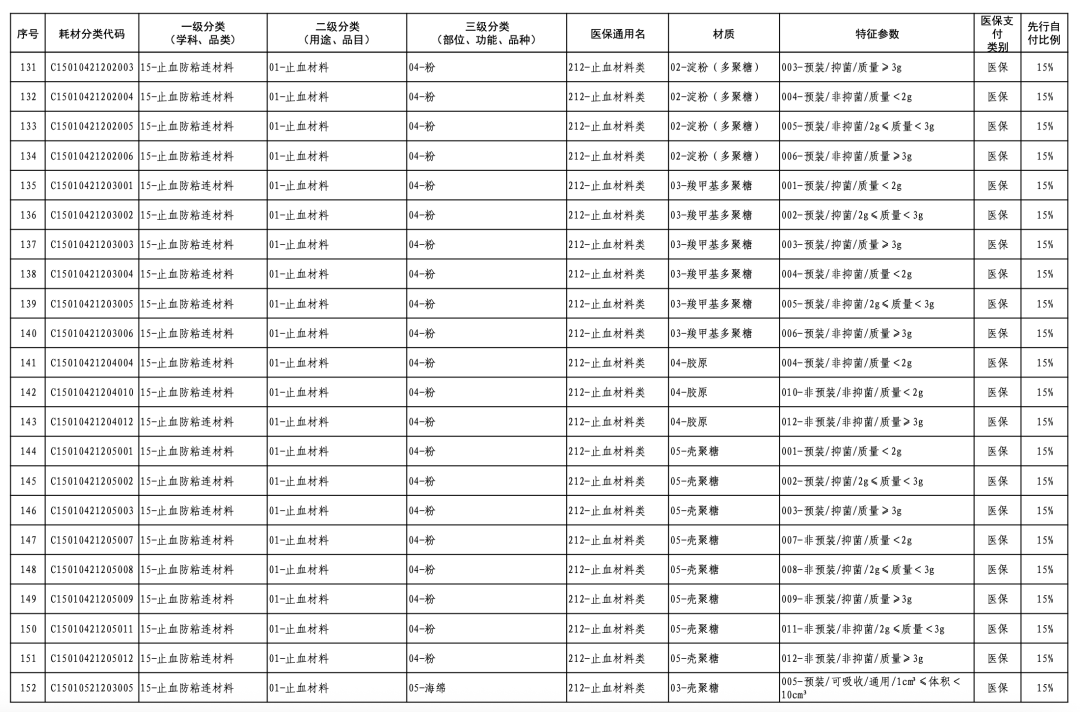
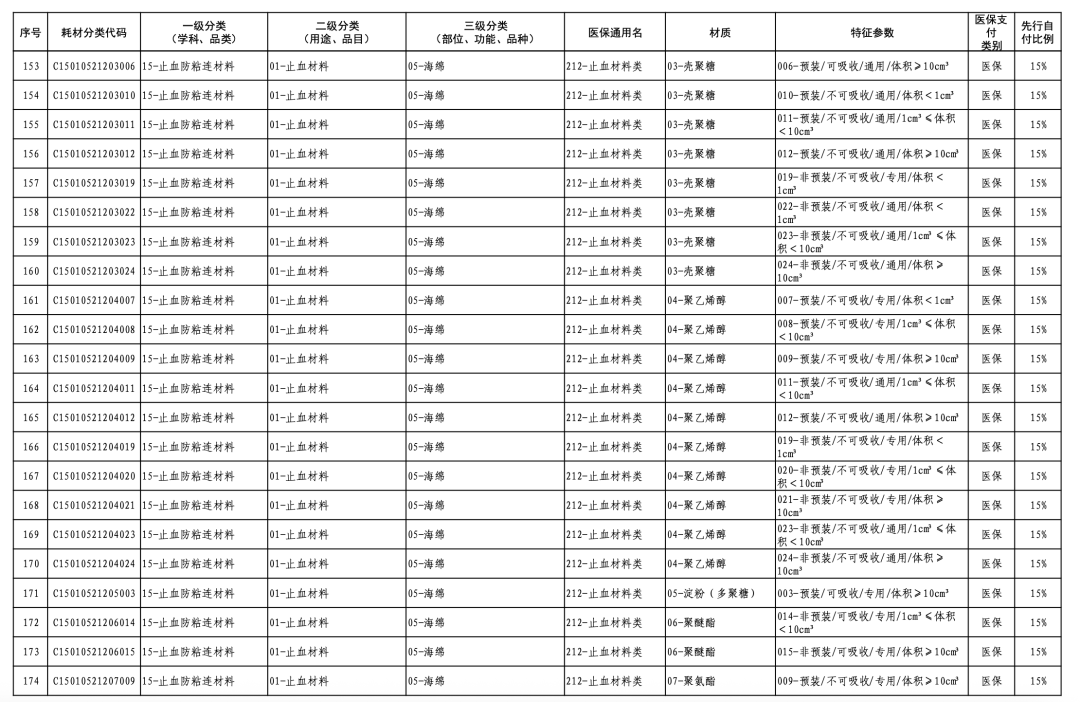
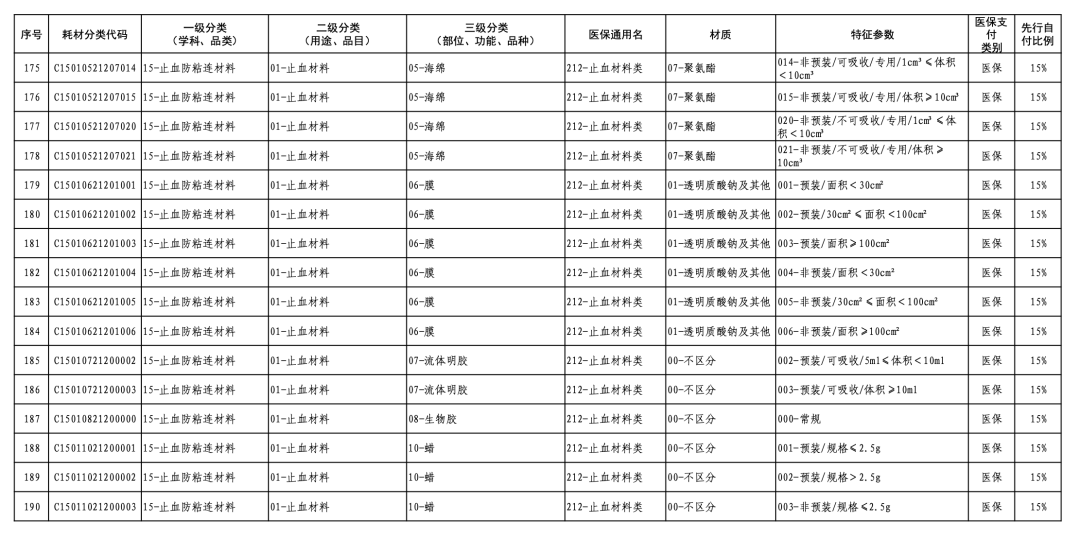
According to the "Notice," a total of 190 new consumables items have been added, covering 18 medical insurance generic name classifications such as 056-Valve Class, 185-Absorbable Suture Class, and 212-Hemostatic Material Class. The adjusted items will be implemented from February 10, 2026.
The Circular also requires healthcare security departments to normalize the supervision of medical consumables usage, with a focus on monitoring items that are frequently used in clinical practice, account for significant fund expenditures, or impose a heavy financial burden on patients.
In January this year, the Fujian Provincial Healthcare Security Administration released the *Trial Directory of Medical Consumables for Medical Insurance Reimbursement in Fujian Province*. The *Directory* implements an admission-based system and will be subject to dynamic adjustments in accordance with regulations. Such adjustments will be based on the operation of Fujian’s medical insurance fund, advancements in medical science and technology, and revisions to medical service pricing projects, while taking into account the needs of insured individuals and clinical practical requirements.
This year, many regions across the country have implemented the new version of the medical insurance consumable catalog and clarified the dynamic adjustment mechanism.
Effective January 1, Shanxi Province will implement the "Shanxi Province Basic Medical Insurance Medical Consumables Catalog (First Batch)," which includes 3,538 Class B items and 248 Class C items, covering materials for vascular/non-vascular interventional therapy, orthopedics, ophthalmology, dentistry, cardiac surgery, and more.
Effective February 1st, the "Henan Province Basic Medical Insurance, Maternity Insurance, and Work-Related Injury Insurance Medical Consumables Catalog (2025)" will be implemented, which includes a total of 4796 consumables, consisting of 4669 general consumables and 127 negotiated consumables.
Effective April 1, the "Liaoning Province Basic Medical Insurance, Work Injury Insurance, and Maternity Insurance Medical Consumables Catalog (2025)" will be implemented, which includes a total of 3388 consumables (67 Class A items and 3321 Class B items).
02
Standardizing Medical Insurance Consumables Payment Management
Transitioning towards generic name management
In recent years, various regions have actively promoted the application of national medical insurance medical consumable codes, leading to coded procurement, coded use, coded settlement, and coded supervision of medical consumables. The catalog admission management of medical consumables is also steadily advancing, with unified basic medical insurance medical consumable catalogs gradually being established within each province, autonomous region, and municipality directly under the central government, and the dynamic adjustment of these consumable catalogs being promoted.
Currently, many regions across the country are further standardizing medical insurance reimbursement management for consumables.
Henan has proposed exploring the establishment of a monitoring mechanism for the procurement and use of medical consumables, monitoring and analyzing aspects such as the payment of medical insurance funds, patient burden, and online procurement, to ensure the stable operation of medical insurance funds.
On January 26, the Tianjin Healthcare Security Administration issued the "Notice of Tianjin Healthcare Security Administration on Doing a Good Job in the Payment Management of Medical Consumables under Basic Medical Insurance (Draft for Comment)". It proposes that the consumable catalog will be managed using "generic name + material characteristics" from the "National Healthcare Security Administration Medical Consumables Classification and Codes", and will gradually transition to management by medical insurance generic name.
For a long time, medical consumable companies have had significant freedom in naming their products, leading to numerous instances of different products sharing the same name and the same product having different names. This makes it difficult to accurately and efficiently identify products, significantly increasing the difficulty of medical insurance and healthcare management.
Currently, the state has released some medical insurance generic names.
In January of this year, the National Healthcare Security Administration (NHSA) released the "Medical Insurance Classification and Codes, and Generic Names for Bone, Cartilage, and Soft Tissue Implants," which, after adjustments, involves 1 primary category, 18 secondary categories, and 126 tertiary categories, resulting in the revision of 260 medical insurance generic names.
This revision embodies three key characteristics. Firstly, it standardizes management through regulated integration and classification. The categories of bone, cartilage, and soft tissue implant consumables are complex, numerous, and feature diverse material characteristics, with the widespread phenomenon of "multiple certificates for one product, multiple items per certificate, and multiple specifications for one item." This classification adjustment optimizes the first-level classification name "Orthopedic Materials" to "Bone, Cartilage, and Soft Tissue Materials," while also integrating relevant materials originally dispersed under neurosurgical materials, dental materials, and repair materials into "Bone, Cartilage, and Soft Tissue Materials."
The revision also gives full consideration to clinical practice. Highlighted the product's value. Select necessary material and characteristic parameters to form the medical insurance generic name, reflecting the clinical value differences of medical consumables. Taking "bone cement" as an example, this revision distinguishes "drug-eluting/non-drug-eluting" as a necessary characteristic of bone cement, while the characteristics of "joint/spine/other sites" are not reflected in the medical insurance generic name.
Furthermore, the revisions, by reasonably setting up classifications, reflect the characteristics of innovative materials in the generic name, supporting the rapid application of innovative products. For example, custom-made implantable materials rely on digital technology and can be customized according to the patient's bone data, making them more individually adaptable than traditional mass-produced medical consumables. Based on this, a new classification of "custom-made implantable materials" was added and the corresponding medical insurance generic name was revised.
According to the National Healthcare Security Administration's plan, for consumable categories with relatively mature generic name management, the state will gradually formulate a nationally unified medical insurance catalog and gradually expand the range of consumable categories covered by the national catalog.
A national unified management system for medical consumables under national medical insurance is taking shape, and the consumables market is poised to enter a more orderly and sustainable phase of healthy development.
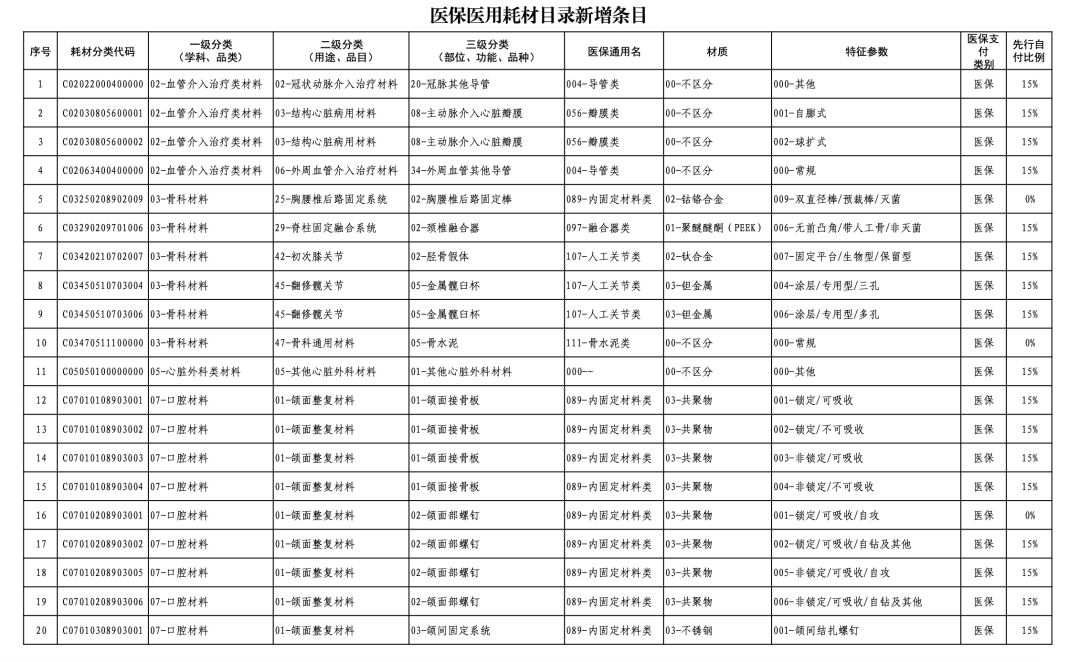
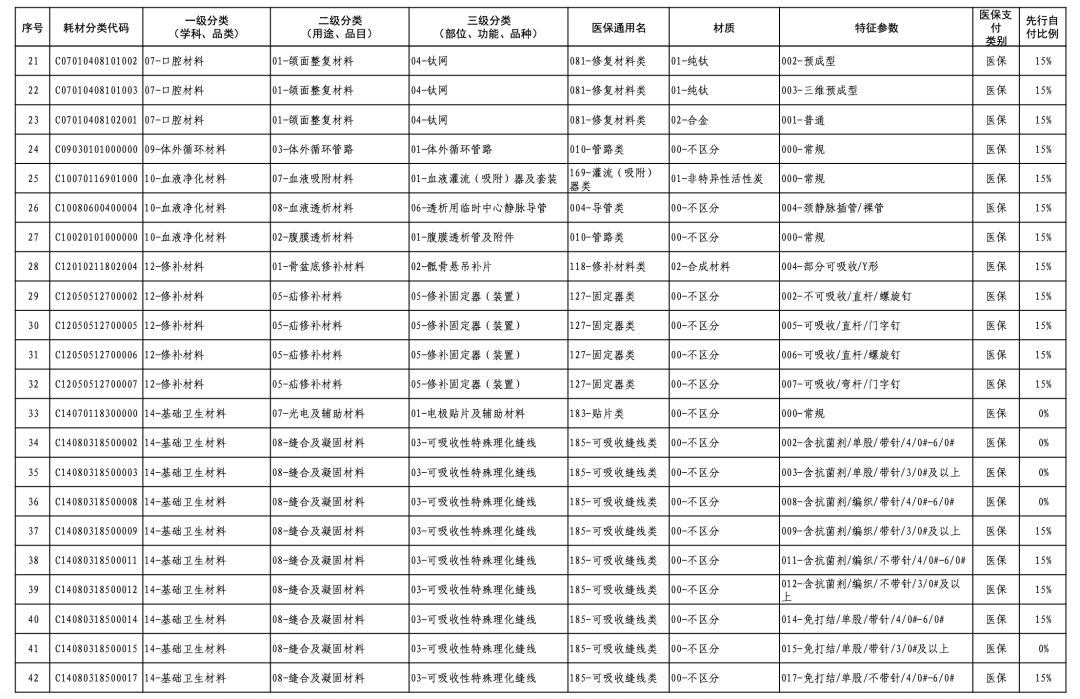
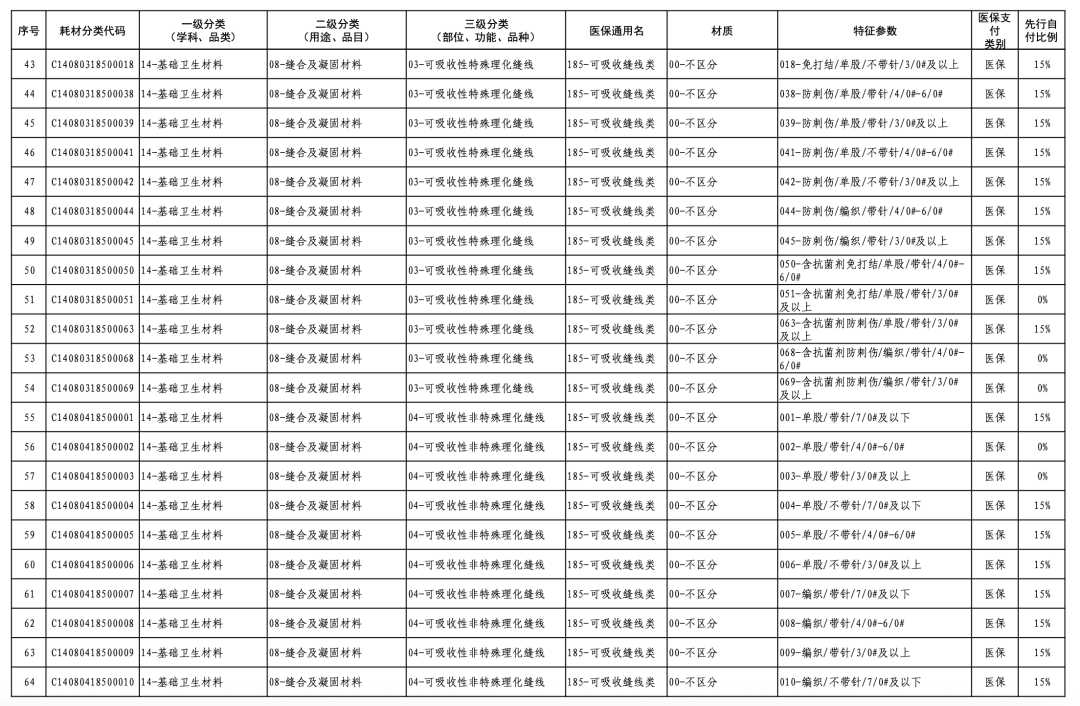
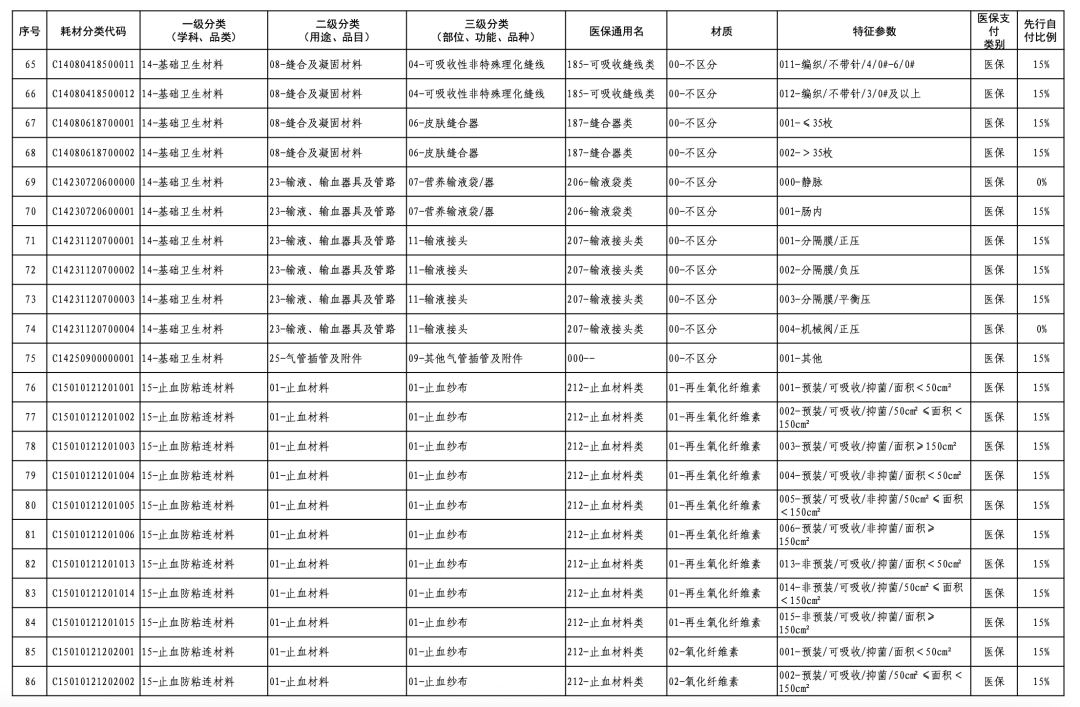
【Copyright and Disclaimer】The above information is collected and organized by PlastMatch. The copyright belongs to the original author. This article is reprinted for the purpose of providing more information, and it does not imply that PlastMatch endorses the views expressed in the article or guarantees its accuracy. If there are any errors in the source attribution or if your legitimate rights have been infringed, please contact us, and we will promptly correct or remove the content. If other media, websites, or individuals use the aforementioned content, they must clearly indicate the original source and origin of the work and assume legal responsibility on their own.
Most Popular
-

Continental Plans to Begin Sale of ContiTech in Early 2026
-

$4 Billion! Medtronic Makes Another Acquisition
-

BASF Delivers First Batch of Innovative Cathode Materials for Semi-Solid-State Batteries to Weilan New Energy
-
Profit and Revenue Growth Struggle to Conceal Debt Repayment Pressure; Success of Kingfa Sci & Tech's High-End Strategy Yet to Be Seen
-

Why did a century-old european dental instrument giant relocate its manufacturing hub to china?








