99.5% selectivity! academician jiang jianchun's team: Solvent-Regulated Precise Catalytic Conversion of HMF
5-Hydroxymethylfurfural (HMF), a biomass platform molecule, is recognized by the U.S. Department of Energy as one of the "Top 10+4" key platform compounds due to its structure containing functional groups such as aldehyde, hydroxymethyl, and furan ring. It shows great potential for conversion into high-value products such as BHMF, DMF, BHMTHF, and 1,6-HDO.
However, the hydrogenation process of HMF has a complex pathway involving multiple intermediates and products. Achieving precise control over the reaction direction in a single catalytic system remains a challenging task.
In response to the challenge of the existing catalytic systems' difficulty in precisely controlling product selectivity, the team led by Academician Jianchun Jiang from the Institute of Chemical Industry of Forest Products, Chinese Academy of Forestry, has innovatively developed a Ni-ZnO/AC coupled solvent multiphase catalytic system using a low-temperature co-precipitation method. This system achieves high selectivity conversion of HMF under a single catalyst system—preferentially producing BHMF in 1,4-dioxane with a selectivity of up to 97.5%, and directionally converting to DMF in isopropanol with a selectivity of up to 99.5%.

The related research results are presented in "A Promising Strategy for Solvent-Regulated Selective Hydrogenation of 5-Hydroxymethylfurfural over Porous Carbon-Supported Ni-ZnO Nanoparticles." In "Nano-Micro Letters" (IF 36.3) 。
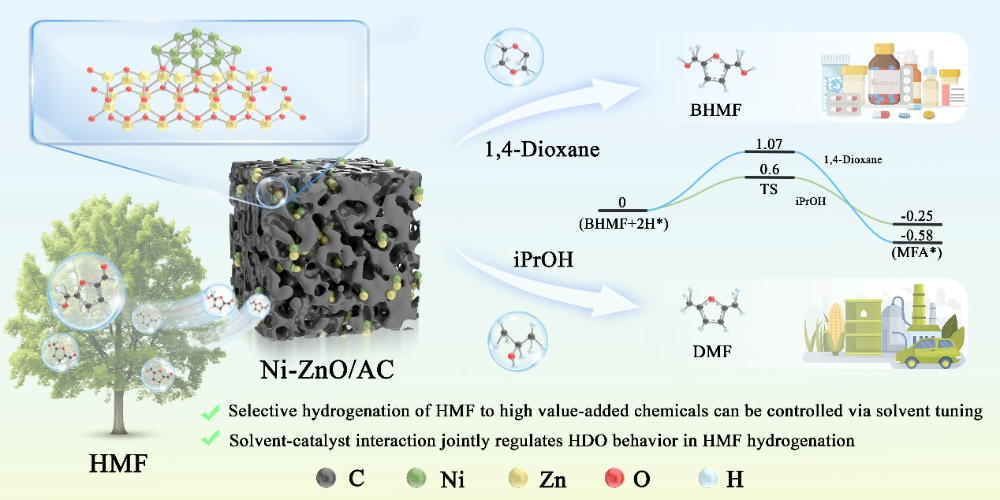
This study systematically reveals for the first time the decisive regulatory effect of solvent polarity and proton donor ability on the product pathway of HMF hydrogenation. The highlights of this paper are as follows:
A composite catalyst of Ni-ZnO nanoparticles supported on coconut shell carbon (Ni-ZnO/AC) was successfully constructed using a low-temperature co-precipitation method, demonstrating excellent catalytic activity and cycle stability in HMF hydrogenation reactions.
By regulating the solvent system, high selective conversion of HMF under a single catalyst system is achieved—preferentially producing BHMF in 1,4-dioxane (with a selectivity of up to 97.5%) and directionally converting to DMF in isopropanol (with a selectivity of up to 99.5%).
The system reveals the synergistic regulation mechanism of reaction pathways by solvent polarity and proton donor ability, proposing a new hydrogenation-deoxygenation strategy based on the hydrogen shuttle effect, providing theoretical support for the precise and high-value utilization of biomass platform compounds.
In this work, dried coconut shells with a particle size of 4–5.6 mm were selected as raw materials, and a porous carbon-loaded Ni-ZnO nanoparticle catalyst (Ni-ZnO/AC) was prepared using a low-temperature co-precipitation method.
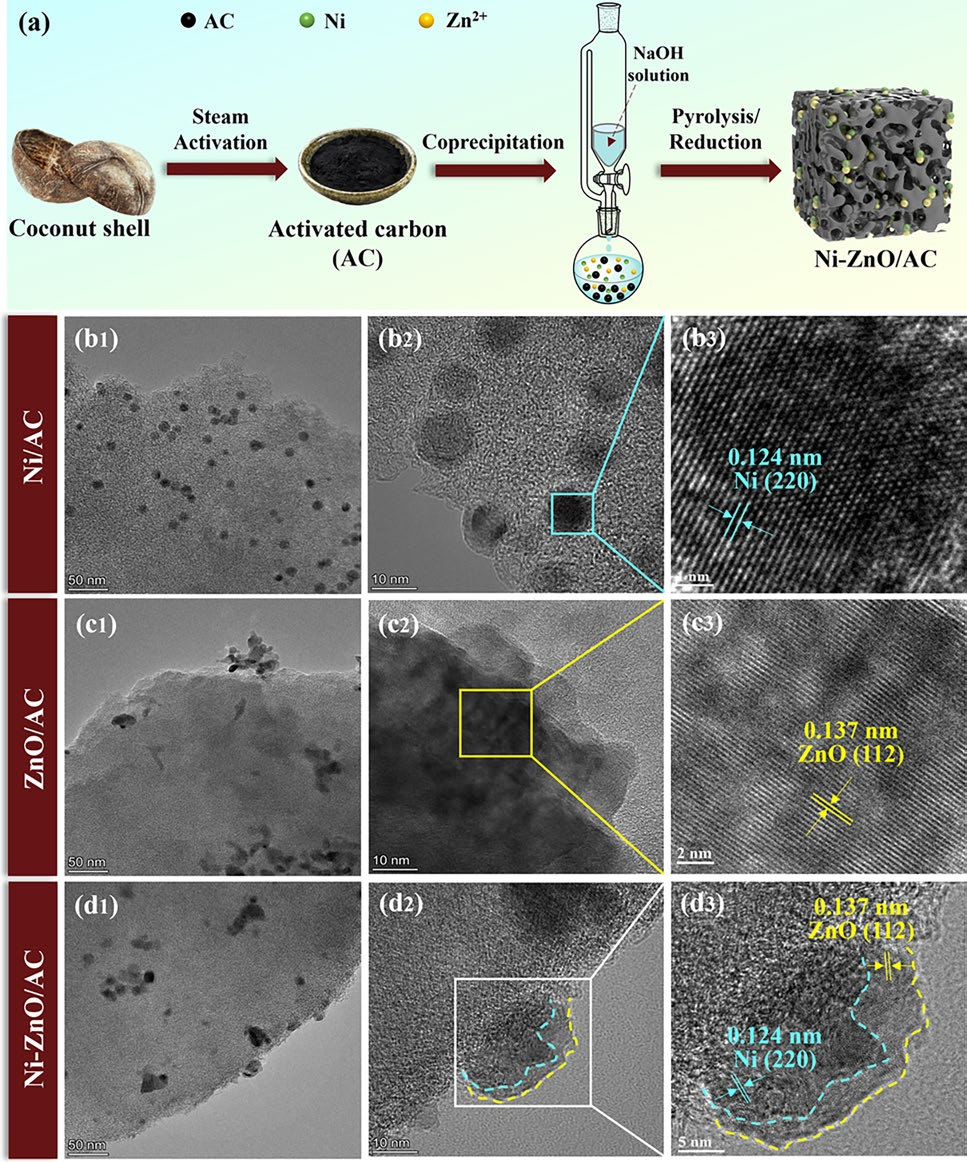
Figure 1 The synthesis process and microstructure characterization of Ni-ZnO/AC. a Synthesis steps of Ni-ZnO/AC. b TEM image of Ni/AC, c TEM image of ZnO/AC, and d TEM image of Ni-ZnO/AC.
02
Compared to 9 types of solvents, the conversion rate of isopropanol reaches 99.5%.
HMF, due to its multi-functional group structure, can undergo hydrogenation reactions in solvents to be converted into various high-value-added compounds.
This study systematically examined the effect of Ni-ZnO/AC catalyst on four types of polar aprotic solvents and five types.Polar protic solventThe effect of hydrogenation reaction on HMF.
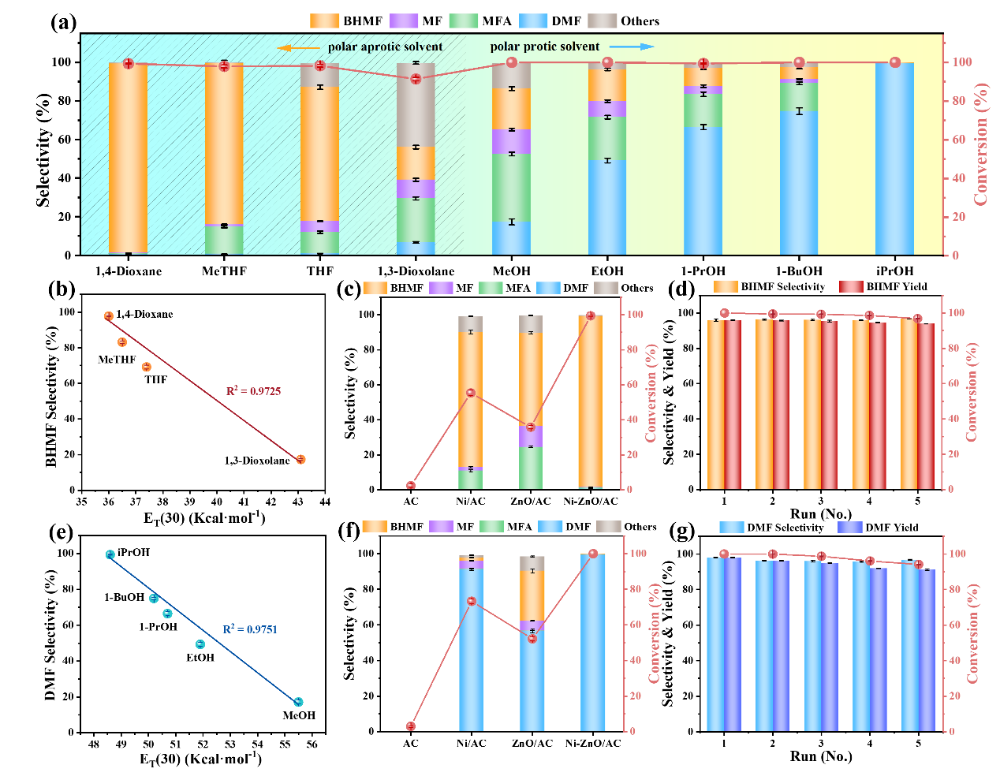
Figure 2The effect of solvent types and polarity on the catalytic hydrogenation of HMF and the recycling performance of Ni-ZnO/AC catalyst
Four types of polar aprotic solvents: 1,4-dioxane, 2-methyltetrahydrofuran, tetrahydrofuran, 1,3-dioxolane.
5 types of polar protic solvents: methanol, ethanol, 1-propanol, 1-butanol, isopropanol
The results show that in polar aprotic solvents, HMF preferentially forms BHMF.2,5-FurandimethanolThe selectivity is as high as 97.5% especially in 1,4-dioxane.
In polar aprotic solvents, it tends to form DMF (2,5-DimethylfuranThe selectivity in isopropanol is as high as 99.5%.
Further analysis revealed that the selectivity of BHMF and DMF is linearly positively correlated with the solvent polarity ET(30) value, indicating that solvent polarity can significantly enhance the solubility of the substrate and the degree of reaction completion, thereby affecting the reaction pathway and product distribution.
In addition, the catalyst maintains good stability in the recycling process within two solvent systems.
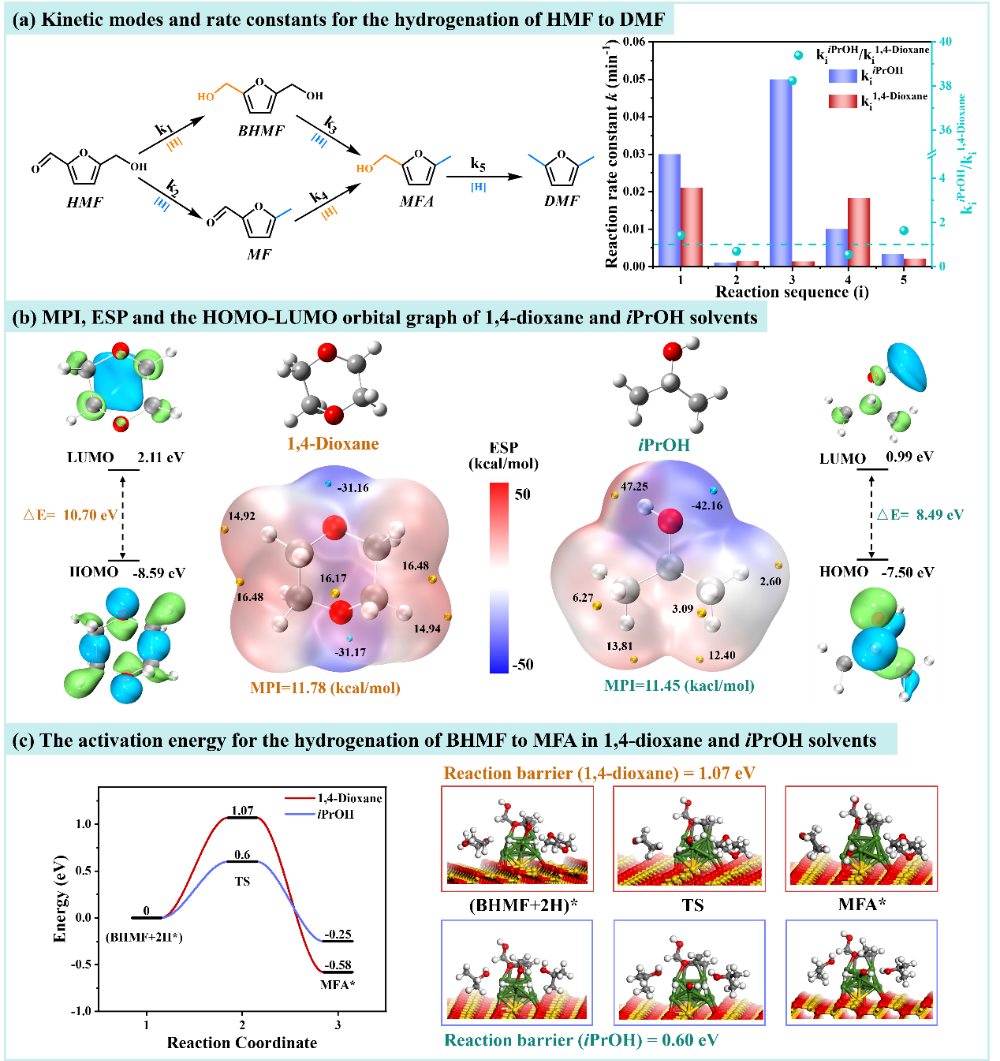
Figure 3 Kinetics, molecular properties, and energy barrier analysis of HMF hydrogenation in 1,4-dioxane/isopropanol solvent.
In isopropanol, the rate constant (k) for the conversion of HMF to BHMF1The rate is 1.5 times higher in 1,4-dioxane, indicating that protic solvents are more favorable for carbonyl hydrogenation under the action of the Ni-ZnO/AC catalyst.
The step of further converting BHMF to MFA (k3In isopropanol, it is 38.2 times that of the latter, indicating that this process is almost inhibited in 1,4-dioxane, BHMF tends to accumulate, and the product selectivity naturally favors BHMF.
Subsequently, by comparing the molecular polarity index (MPI), electrostatic potential distribution (ESP), and HOMO-LUMO energy gap (ΔE) of the solvents, it was found that although 1,4-dioxane has a slightly higher polarity, it lacks proton donor capability, making it difficult to facilitate the crucial proton transfer.
Isopropanol not only has a smaller ΔE and stronger charge polarization capability, but it can also directly provide protons and stabilize reaction intermediates, offering strong support for subsequent dehydroxylation and hydrogenation processes.
Furthermore, DFT calculations based on the explicit solvent model reveal that the hydrogenolysis reaction of BHMF on the Ni-ZnO/AC surface has a barrier of only 0.60 eV in an isopropanol system, which is significantly lower than the 1.07 eV in a 1,4-dioxane system.
Isopropanol participates in the reaction through the "hydrogen shuttle effect," not only does its proton accelerate the departure of the hydroxyl group, iPrO...-It can also capture active hydrogen on the Ni surface, achieve solvent self-regeneration, and complete an integrated cycle.
Although solvent polarity has some influence on the regulation of reaction products, it is the proton donor ability that is the key factor determining the reaction pathway and product selectivity.
Polar protic solvents represented by isopropanol not only enhance the solubility of substrates and the contact with catalysts but also actively participate in the reaction through hydrogen bonding and proton transfer processes, greatly promoting the highly selective conversion of HMF to DMF.
Summary and Outlook
This study constructed a Ni-ZnO/AC heterogeneous catalyst coupled with different solvent catalytic conversion systems, achieving precise conversion of the biomass platform molecule HMF to high value-added products.
In 1,4-dioxane/isopropanol, BHMF and DMF are generated with high selectivity, respectively.
The study, combining experimental results and DFT calculations, found that the solvent not only serves as a reaction medium but also forms a synergistic effect with the catalyst, profoundly influencing the reaction rate and path selection.
This strategy effectively manipulates product distribution by regulating solvent polarity and hydrogen-donating ability, providing a controllable new approach for green chemical transformations.
This research work was supported by the National Natural Science Foundation of China's Outstanding Youth Fund (32222058) and the Fundamental Research Funds of CAF (CAFYBB2022QB001). Huang Rulu, a doctoral student from the 2022 class at the Institute of Chemical Industry of Forest Products, is the first author. Academician Jiang Jianchun and Researcher Wang Kui from the Institute of Chemical Industry of Forest Products, Researcher Tian Ziqi from the Ningbo Institute of Materials Technology and Engineering, Chinese Academy of Sciences, and Professor Chai Yongming from China University of Petroleum (East China) are the co-corresponding authors.
【Copyright and Disclaimer】The above information is collected and organized by PlastMatch. The copyright belongs to the original author. This article is reprinted for the purpose of providing more information, and it does not imply that PlastMatch endorses the views expressed in the article or guarantees its accuracy. If there are any errors in the source attribution or if your legitimate rights have been infringed, please contact us, and we will promptly correct or remove the content. If other media, websites, or individuals use the aforementioned content, they must clearly indicate the original source and origin of the work and assume legal responsibility on their own.
Most Popular
-

A Look at the Material Suppliers Behind SpaceX
-

BASF, Selling Again!
-

BASF Cuts Core Business As It Exits All Non-Core Operations, Triggering A New Round Of Industry Restructuring
-

Gas Explosion Accident at Shanxi Tongzhou Group Liushenyu Coal Mine Results in 82 Deaths
-

Bola Carbon Black Announces Global Restructuring of Specialty Carbon Black and Multi-Walled Carbon Nanotube Business







