Major announcement: Nearly $300 Million Medical Equipment Procurement Contract Awarded, Policy Benefits Continue to Be Released
Recently, a batch of medical equipment renewal projects announced on the China Government Procurement Network, with a total budget of nearly 300 million yuan, covers a rich variety of popular products such as 256-slice CT, fully automatic biochemical analyzers, and color Doppler ultrasound, injecting strong momentum into the 2026 medical equipment market. This signal clearly indicates that the wave of medical equipment renewal has smoothly continued into the new year, and the industry is welcoming an unprecedented window of development. At the same time, the falling of the "heavy hammer" of regulation is also reshaping the competitive landscape of the market.
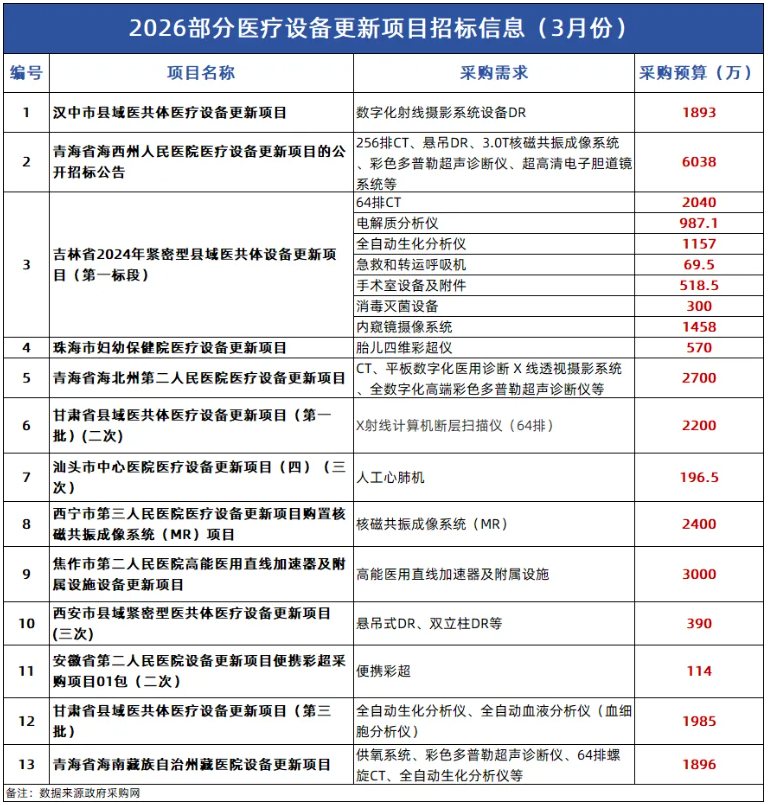
Policy and funding drive the continuous release of medical equipment market demand.
The anticipated boom in the medical device market in 2026 stems from precise top-level planning and sustained financial support. As a key policy guideline for the healthcare sector, the Central Government’s No. 1 Document of 2026 explicitly calls for upgrading county-level hospitals and key central township health centers. Furthermore, the “Implementation Plan for the Healthcare Infrastructure Strengthening Project” specifies that, during the 15th Five-Year Plan period, approximately 1,000 tightly integrated county-level medical consortia will be supported, with a focus on improving infrastructure and equipment in weak departments of county-level public hospitals and in township health centers.
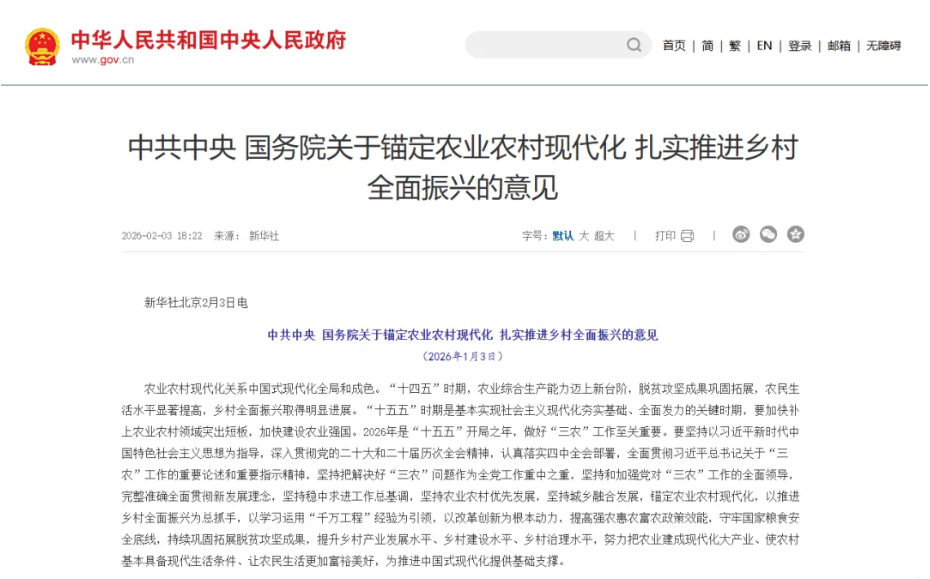
This policy direction has not only driven the upgrade and iteration of high-end, intelligent medical devices but also continuously unleashed procurement demand in county-level and grassroots markets—from smart equipment upgrades in developed eastern counties and basic diagnostic equipment supplementation in central and western counties, to the enhancement of emergency and critical care equipment in remote mountainous areas—making the grassroots market a new engine for growth in the medical device sector.
The implementation of financial support has provided a solid foundation for the renewal of medical equipment. In January this year, the National Development and Reform Commission issued a document stating that the first batch of 93.6 billion yuan in ultra-long-term special treasury bonds for equipment renewal in 2026 has been officially allocated, with the medical field listed as a key support area. The injection of these funds has effectively alleviated the financial pressure on medical institutions at all levels, especially grassroots medical institutions, for equipment procurement, facilitating the transformation of equipment renewal from "intention" to "action." At the same time, the coordinated efforts of multi-channel funding have further reduced the procurement costs for medical institutions, also providing a broader market space for domestic medical device enterprises.
II. Continued tightening of regulation to standardize market competition order
As the market accelerates in, regulatory efforts are also intensifying, with "strict regulation and promotion of standardization" becoming the main theme of industry development. A series of regulatory measures are being implemented, breaking industry chaos and pushing the market back to rational competition.
The Ministry of Finance took the lead in cracking down on abnormal low prices. Starting from February 1st, new regulations targeting abnormal low prices in medical equipment procurement came into effect, clearly stating that hospitals will no longer solely focus on price. "Total lifecycle cost" and "technical quality score" have become the core factors for winning bids. This regulation has completely broken the previous model where some companies used "low-price bidding" and then made profits through consumables. It now forces companies to return to the product itself, focusing on technology, quality, and long-term service.
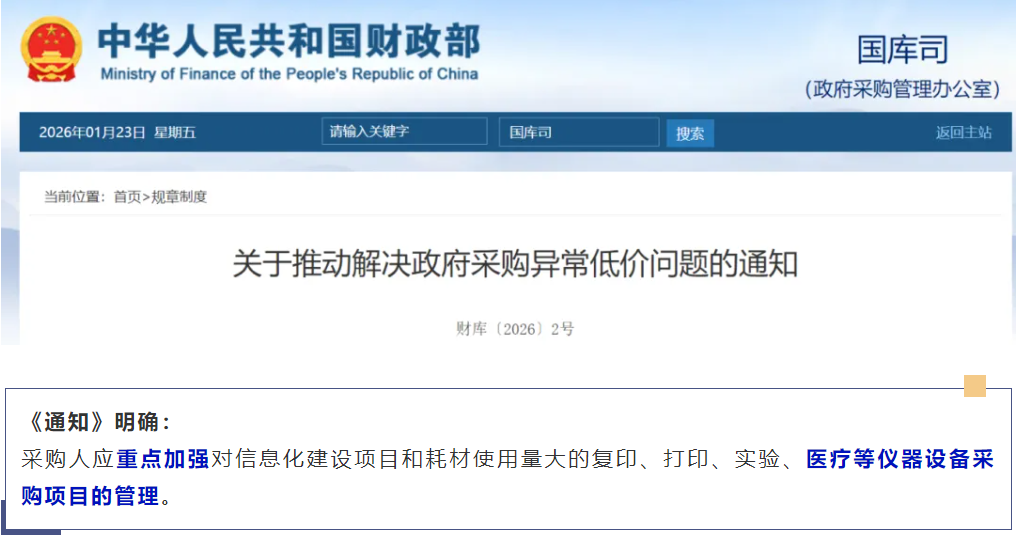
At the same time, the National Development and Reform Commission has explicitly proposed to address the "involutionary" competition in the medical equipment procurement sector, continuously increasing the weight of "service score" and "quality and brand score" in the centralized procurement documents, making technical parameters not just a formality but a "hard currency" for measuring the competitiveness of enterprises. In addition, the Central Commission for Discipline Inspection has named "technical backdoors" in medical equipment procurement, incorporating exclusive and biased parameter settings into the "negative list for procurement," making hidden parameters an untouchable "audit red line," effectively standardizing the procurement process and maintaining a fair and competitive market order.
III. Regulatory Oversight Hides Opportunities—Dealers Enter a Critical Transformation Period
What appears to be tightening regulation is, in fact, a catalyst for high-quality industry development, opening a new door of opportunity for medical device distributors. Stricter oversight is compelling hospital equipment departments to strike a balance between "compliance" and "practicality"—they must establish rigorous procurement specifications that can withstand audits to avoid compliance violations, while also ensuring the acquired equipment meets clinical needs, avoids idle waste, and genuinely enhances diagnostic and treatment capabilities.
For medical device distributors, transformation has become an inevitable choice: the traditional model of "selling equipment without providing solutions" is no longer sustainable. The market demand is shifting from "product supply" to "value-added services." Whoever can precisely grasp the core pain points of hospitals and help them solve the difficulties of "technical parameter validation" and "configuration rationality" will be able to transform from a mere "equipment seller" into a trusted "equipment planning consultant" for hospitals.
This transformation requires distributors not only to possess solid product knowledge but also to have professional technical service capabilities—whether providing hospitals with full lifecycle cost analyses of equipment, developing customized equipment configuration plans based on regional disease profiles and clinical needs, or assisting hospitals in procurement specification validation and compliance risk mitigation—all of which will become core competitive advantages for distributors.
IV. Conclusion: Seizing Opportunities and Embarking Together on a New Journey toward High-Quality Development in the Medical Device Industry
In 2026, the medical equipment market is at a critical juncture where policy benefits and regulatory standards coexist. The dual drive of policies and funds continuously injects vitality into the market; while the continuous tightening of regulations pushes the industry to bid farewell to "unrestrained growth" and enter a new stage of standardized, high-quality development.
For medical device companies and distributors, only by closely following policy guidance, adhering to the bottom line of compliance, focusing on technological innovation and service upgrades, and proactively transforming to meet market demands, can they stand out in a market environment full of both opportunities and challenges, share in the dividends of industry development, and contribute to the improvement and upgrading of our country's medical and health care industry.
【Copyright and Disclaimer】This article is the property of PlastMatch. For business cooperation, media interviews, article reprints, or suggestions, please call the PlastMatch customer service hotline at +86-18030158354 or via email at service@zhuansushijie.com. The information and data provided by PlastMatch are for reference only and do not constitute direct advice for client decision-making. Any decisions made by clients based on such information and data, and all resulting direct or indirect losses and legal consequences, shall be borne by the clients themselves and are unrelated to PlastMatch. Unauthorized reprinting is strictly prohibited.
Most Popular
-

Not Just MDI! Huntsman and BASF Impose “War Surcharge” Across Europe
-

Middle East Triggers Widespread Force Majeure In Global Chemical Industry
-

BASF Delivers First Batch of Innovative Cathode Materials for Semi-Solid-State Batteries to Weilan New Energy
-

Deadly Impact: Hormuz Strait Blockade Sparks Shortage of Plastic Raw Materials, Threatening Shutdowns at Japanese and Korean Chemical Plants
-

BASF Leads The Surge! Additive Costs Soar Across The Board, How Should Plastics Enterprises Respond?







