Tokyo Science University Develops Low-Temperature Hydrogen Battery to Overcome Hydrogen Storage Barriers
According to foreign media reports, researchers at the Institute of Science Tokyo in Japan have developed a hydrogen battery that operates at a temperature of only 90°C, overcoming the limitations of high temperature and low capacity associated with previous methods.
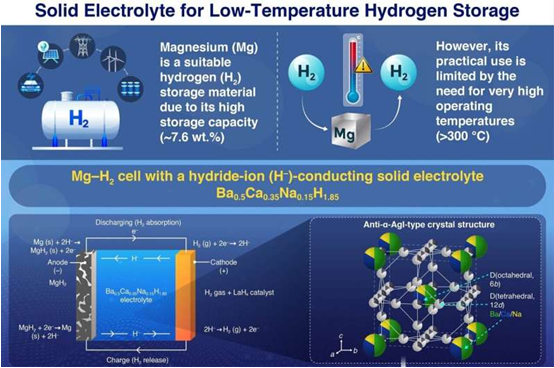
Image source: Tokyo University of Science
The working principle of this device is the movement of hydrogen anions in a solid electrolyte, allowing magnesium hydride, as the anode, to repeatedly store and release its full capacity of hydrogen. This battery provides a practical method for hydrogen fuel storage, paving the way for hydrogen-powered vehicles and clean energy systems.
One of the most pressing challenges facing hydrogen use is its storage, which often requires very low temperatures (-252.8°C) and high pressures (350 to 700 bar). A more effective approach than storing hydrogen in gas or liquid form is to store it in solid materials with high theoretical storage capacities, such as magnesium hydride (MgH2).
This material can be integrated into battery-like systems, where not only mobile electrons, but also hydrogen itself can be stored and released during the charging and discharging processes.
Until recently, this method was still limited by the following: the need for high operating temperatures above 300°C, poor reversibility of hydrogen absorption and desorption, and adverse side reactions that reduce performance.
The relevant paper of this study was published in the journal "Science," and was completed by the research team led by Dr. Takashi Hirose, Associate Professor Naoki Matsui, and Professor Ryoji Kanno from the Research Center for All-Solid-State Battery, Institute of Science, Tokyo.
Matsui stated, "We demonstrated the performance of Mg–H2 batteries as safe and efficient hydrogen energy storage devices, achieving high capacity, low temperature, and reversible hydrogen absorption and release."
The innovation of this battery lies in its solid electrolyte Ba0.5Ca0.35Na0.15H1.85, which can efficiently transport hydrogen ions, especially hydride ions (H-). This material has a reverse α-AgI-type crystal structure, renowned for its exceptionally high ionic conductivity. In this structure, barium, calcium, and sodium occupy the body-centered positions, while H- moves through face-sharing tetrahedral and octahedral interstitial sites, enabling free migration. Tests indicate that this material has a high ionic conductivity (2.1 × 10^-5 S cm^-1) and electrochemical stability at room temperature, allowing the system to effectively store and release hydrogen gas over an extended period.
The battery design uses MgH2 as the anode and hydrogen gas (H2) as the cathode. During charging, MgH2 releases H–, which migrates through the Ba0.5Ca0.35Na0.15H1.85 electrolyte to the H2 cathode, where it is oxidized to release H2 gas. During discharging, the opposite occurs: H2 gas at the cathode is reduced to H–, which migrates through the electrolyte to the anode and reacts with Mg to form MgH2. This process allows the battery to store and release hydrogen gas as needed, with all processes occurring at controlled temperatures below 100°C. Using this battery, researchers were able to achieve the full theoretical storage capacity of MgH2 in repeated cycles, which is approximately 2,030 mAh g-1, equivalent to 7.6 wt.% (weight percent) of hydrogen gas.
Traditional solid-state hydrogen storage methods face significant limitations. The thermally driven absorption and desorption process requires extremely high temperatures of 300 to 400°C to achieve hydrogen release or capture, making the process energy-intensive and unsuitable for everyday applications. Another method uses liquid electrolytes for electrochemical hydrogen storage at lower temperatures, but the hydrogen ion transport performance is poor, meaning these materials cannot achieve levels close to their theoretical hydrogen storage capacity. Therefore, neither method has provided an efficient, reversible, and low-temperature hydrogen storage solution.
Hirose explained, "These characteristics of our hydrogen storage batteries were previously unattainable by traditional thermal methods or liquid electrolytes, laying the foundation for constructing an efficient hydrogen storage system suitable as a hydrogen energy carrier."
This type of battery could be key to the future of hydrogen power, driving the development of hydrogen-powered vehicles and zero-carbon emission industries.
【Copyright and Disclaimer】The above information is collected and organized by PlastMatch. The copyright belongs to the original author. This article is reprinted for the purpose of providing more information, and it does not imply that PlastMatch endorses the views expressed in the article or guarantees its accuracy. If there are any errors in the source attribution or if your legitimate rights have been infringed, please contact us, and we will promptly correct or remove the content. If other media, websites, or individuals use the aforementioned content, they must clearly indicate the original source and origin of the work and assume legal responsibility on their own.
Most Popular
-

A Look at the Material Suppliers Behind SpaceX
-

Two Major Chemical Giants to Shut Down, Sell Again
-

BASF, Selling Again!
-

BASF Cuts Core Business As It Exits All Non-Core Operations, Triggering A New Round Of Industry Restructuring
-

Gas Explosion Accident at Shanxi Tongzhou Group Liushenyu Coal Mine Results in 82 Deaths







