Efficiently Recycle PET Using Air Moisture, with an Efficiency Rate of Up to 94%
Polyethylene terephthalate (PET) is one of the most widely used plastics, crucial in beverage bottles, food packaging, and textiles. With an annual global production approaching 70 million tons, PET accounts for 67% of the market share in the beverage industry. However, excessive use and low recycling rates mean that PET makes up 12% of solid waste.
A research team from Northwestern University in the United States has developed a groundbreaking method to break down PET without using toxic chemicals, excessive heat, or harsh solvents. Their approach utilizes trace amounts of moisture in the air to selectively decompose PET into its original monomers, which can then be recycled into new plastics or even upgraded into higher-value materials.
"America is the country with the most severe per capita plastic pollution, and we only recycle 5% of our plastics," said Yosi Kratish, the lead researcher on the project. "There is an urgent need for better technologies to handle different types of plastic waste. Most methods today involve melting plastic bottles and then downcycling them into lower quality products. What is particularly exciting about our research is that we use moisture from the air to break down plastics, achieving an exceptionally clean and selective process."
The new process uses a simple, abundant molybdenum catalyst with activated carbon as the carrier. The PET plastic is exposed to this catalyst and heated to 250-265°C, which is just slightly above the melting point of PET. Once the chemical bonds of the plastic are weakened, exposure to air completes the decomposition process, converting PET into terephthalic acid (TPA), a valuable chemical used for making new plastics. Unlike traditional methods, this process produces almost no waste byproducts—only acetaldehyde, an industrial chemical that is easy to remove.
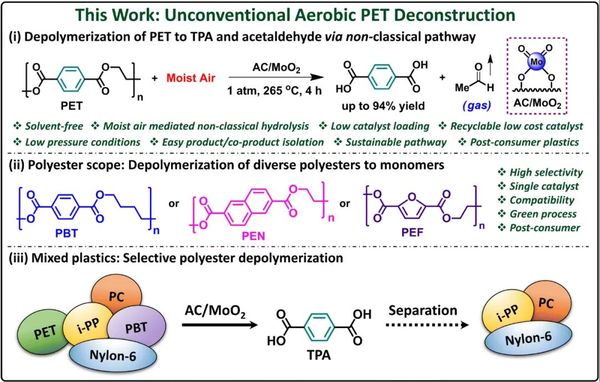
This method has several advantages over existing recycling technologies. First, it eliminates the need for expensive and toxic solvents, reducing environmental impact. The reaction is also highly selective, meaning it only breaks down PET without affecting other plastics. This eliminates the costly and time-consuming need to sort plastic waste before recycling.
"There are many drawbacks to using solvents," Klatish explained. "They can be expensive, and you have to heat them to high temperatures. Then, after the reaction, you end up with a bunch of materials that must be sorted to recover the monomers. Instead of using solvents, we used water vapor from the air. This is a more elegant way to solve the problem of plastic recycling."
The speed of the process is another major advantage. Traditional chemical recycling methods may take up to 72 hours, while Northwestern University's method can nearly completely decompose PET in just four hours. The catalyst itself is also durable and reusable, maintaining its effectiveness over multiple recycling cycles.

The photo shows the formation of TPA products after each 4-hour heating in different runs (total reaction time = 20 hours).
"The results show that the process is fast and effective," said Naveen Malik, the first author of the study. "After just four hours, 94% of TPA was recovered. The catalyst is durable and recyclable, meaning it can be used over and over again without losing its efficacy."
In addition, the method is applicable to real-world plastic waste. Researchers tested it on plastic bottles, polyester fibers, and even mixed plastic waste, finding that it successfully extracted pure, colorless TPA from colored plastics. Unlike other methods for PET that require pre-cleaning, this process works even on contaminated plastic waste.
The Northwestern University team is now working to scale up the process to handle industrial-scale plastic recycling. With further optimization, they believe this technology can significantly reduce global plastic pollution and contribute to the development of a circular economy, allowing plastics to be continuously reused rather than discarded.

Aerobic solvent-free AC/MoO2-mediated degradation of post-consumer plastic materials A-D. Reaction conditions: 100 mL Schlenk flask, post-consumer polyester stored in air, AC/MoO2 (3.24 wt% Mo), solvent-free, 265 °C, air. Yield determined by 1H NMR using m-xylene as an internal standard.
"Our technology has the potential to significantly reduce plastic pollution, lower the environmental footprint of plastics, and contribute to the development of a circular economy, allowing materials to be reused rather than discarded," Malik said. "This is a tangible step towards a cleaner, greener future, demonstrating how innovative chemistry can address global challenges in a way that is in harmony with nature."
If adopted on a large scale, this technology can help address one of the world's most pressing environmental challenges—plastic waste. By eliminating the need for harsh chemicals and excess energy, this method makes recycling more practical, cost-effective, and environmentally friendly.
As millions of tons of plastic waste continue to accumulate each year, such innovations can play a key role in building a more sustainable future.
【Copyright and Disclaimer】The above information is collected and organized by PlastMatch. The copyright belongs to the original author. This article is reprinted for the purpose of providing more information, and it does not imply that PlastMatch endorses the views expressed in the article or guarantees its accuracy. If there are any errors in the source attribution or if your legitimate rights have been infringed, please contact us, and we will promptly correct or remove the content. If other media, websites, or individuals use the aforementioned content, they must clearly indicate the original source and origin of the work and assume legal responsibility on their own.
Most Popular
-

A Look at the Material Suppliers Behind SpaceX
-

Two Major Chemical Giants to Shut Down, Sell Again
-

BASF, Selling Again!
-

BASF Cuts Core Business As It Exits All Non-Core Operations, Triggering A New Round Of Industry Restructuring
-

Gas Explosion Accident at Shanxi Tongzhou Group Liushenyu Coal Mine Results in 82 Deaths







