New Hope for Plastic Pollution Control: Tokyo University of Science R&D of Heat-Resistant Enzyme Marks Breakthrough in PET Bio-Recycling
Plastic waste continues to pose a serious challenge to the environment, and a study by Tokyo University of Science (TUS) has brought hope for sustainable solutions. Researchers explored bioplastic recycling technology (biorecycling), which uses enzymes or microorganisms to break down polymer molecules.
Among numerous highly promising research subjects, microbial cutinases stand out—they are naturally produced by bacteria and fungi to degrade plant cuticles and have now demonstrated potential for recycling polyethylene terephthalate (PET), a common plastic widely used in plastic bottles and synthetic fibers.

This study was led by Dr. Tatsuya Nishino and Assistant Professor Sho Ito of Tokyo University of Science, in collaboration with graduate students Ryohei Nojima and Lirong Chen. The research focused on the thermostable cutinase CtCut produced by the thermophilic fungus Chaetomium thermophilum. The team analyzed this enzyme under high-temperature conditions required for PET recycling, and their findings—revealing the intrinsic mechanisms underlying CtCut’s structural stability and catalytic activity at industrially relevant temperatures—were recently published in the journal Crystals.
“In recent years, plastic waste has become a prominent environmental issue, making the development of environmentally friendly recycling technologies urgently needed,” said Tatsuhito Nishino. “Therefore, we aim to elucidate the molecular mechanisms of enzymes that remain functional at high temperatures, thereby supporting the development of practical recycling technologies.”
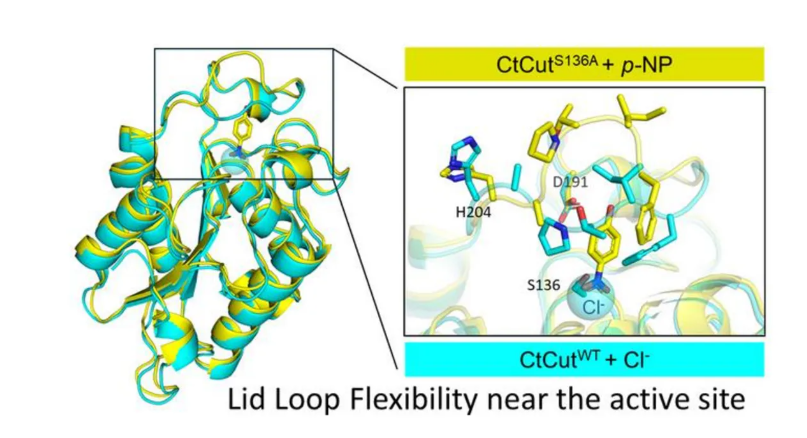
Keratinase possesses a rigid α/β hydrolase core structure, and a flexible lid loop is present near the active site. Structural comparisons show that the lid loop is more prone to conformational changes compared to the rigid core. (Image source: Professor Tatsuya Nishino, Tokyo University of Science)
Balancing Stability and Flexibility: The Core of Enzyme Molecular Design
PET recycling achieves maximum efficiency at approximately 70°C, where the plastic becomes more flexible and easier to process. However, enzymes used for recycling must retain structural integrity at elevated temperatures while simultaneously maintaining sufficient flexibility at their active sites to enable molecular recognition and catalytic reactions. Structural rigidity and flexibility are typically mutually exclusive, making this dual requirement a major challenge in enzyme engineering.
To this end, the research team constructed various CtCut enzyme variants, including wild-type (CtCutWT) and mutant (CtCutS136A) — the latter replacing serine at position 136 with alanine. The team used differential scanning calorimetry to analyze the thermal stability of the enzyme within a temperature range of 30°C to 100°C.
Structurally, CtCut adopts a highly stable α/β hydrolase fold, which is typical for cutinases; its active site is covered by a flexible cap loop, which can open and close during catalysis. Notably, even in the absence of a substrate, chloride ions can still be detected near the active site, indicating the formation of a positive electrostatic microenvironment that may facilitate ligand binding.
During the heating process, the enzyme exhibits a two-step unfolding process, with structural transitions beginning at 60°C and peaking in the range of 65–70°C. This indicates that there are differences in the thermal stability of different regions of the enzyme molecule, suggesting that the protein has structurally distinct domains.
"Research results indicate that there may be functional compartments within enzymes. We observed that dynamic regions near the active site undergo structural changes upon ligand binding, and thermal denaturation occurs in a multi-step process," said Hideto Nishino.
The Significant Importance of Sustainable Plastic Recycling
This study highlights the crucial role of balancing stability and flexibility in enzyme molecular design: a rigid core provides the necessary thermal stability for industrial applications, while a flexible lid loop allows the enzyme to adapt to substrate molecules, enhancing catalytic efficiency.
"This research can provide a design basis for engineering enzymes that possess both heat resistance and the ability to catalyze polymer degradation. In the future, it is expected to promote the implementation of efficient PET decomposition and recycling technologies, thereby addressing the increasingly serious problem of plastic pollution and contributing to the construction of a sustainable, resource-circulating society," said Professor Nishino.
By deepening the understanding of the structure and function of heat-resistant enzymes, this research lays the foundation for innovative recycling technologies, which are expected to significantly reduce plastic waste and promote environmental sustainability. As the global demand for efficient recycling solutions continues to grow, biorecycling technology is becoming an important pathway towards a cleaner and greener future.
Independent research at Los Alamos National Laboratory, USA
At the end of last year, another independent study from Los Alamos National Laboratory also explored the advancements in enzyme engineering technology to improve the efficiency of PET plastic degradation. The research, led by Chief Investigator Thomas M. Groseclose from the laboratory's Bioscience Division, was assisted by Hau B. Nguyen.
The research team pointed out that over the past two decades, scientists have discovered various natural enzymes capable of degrading PET, providing a promising new pathway for recycling this ubiquitous plastic. However, these enzymes still require further optimization to achieve efficient and cost-effective industrial application.
Current research on engineering PET-degrading enzymes primarily focuses on rational design and semi-rational design, while integrating emerging technologies such as directed evolution, high-throughput screening, and computational design. These advances aim to enhance enzyme performance and scalability potential, paving the way for the practical implementation of sustainable PET recycling solutions.
“Although dozens of enzyme engineering studies have already accumulated rich data, future high-throughput screening and directed evolution studies will generate massive amounts of random and combinatorial mutational data across diverse enzyme scaffolds, continuously enhancing the value and richness of such data,” summarized the Los Alamos team.
【Copyright and Disclaimer】This article is the property of PlastMatch. For business cooperation, media interviews, article reprints, or suggestions, please call the PlastMatch customer service hotline at +86-18030158354 or via email at service@zhuansushijie.com. The information and data provided by PlastMatch are for reference only and do not constitute direct advice for client decision-making. Any decisions made by clients based on such information and data, and all resulting direct or indirect losses and legal consequences, shall be borne by the clients themselves and are unrelated to PlastMatch. Unauthorized reprinting is strictly prohibited.
Most Popular
-
From Compliance to Competitiveness: Alpla’s Circular Design and Recycling Practices Driven by PPWR
-

Overseas News: PPG Announces Global 20% Price Increase; Sika Launches High-Performance Polyamide (PA) New Product; AIP Announces Nearly $10 Billion Acquisition
-

15 Companies, 30 Material Technologies: 2026 Rubber & Plastics Exhibition Visitor Guide (I)
-

Driving Intelligent Upgrades In Rubber And Plastics Industry! Preview Of Concurrent Activities At Chinaplas 2026 International Rubber & Plastics Exhibition (Part 2)
-

BASF Delivers First Batch of Innovative Cathode Materials for Semi-Solid-State Batteries to Weilan New Energy








